Rare Disease Genetic Testing Market Outlook:
Rare Disease Genetic Testing Market size was valued at USD 1.29 billion in 2025 and is set to exceed USD 5.36 billion by 2035, registering over 15.3% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of rare disease genetic testing is estimated at USD 1.47 billion.

The increasing prevalence of rare diseases, advancements in genomic technologies, and the growing adoption of personalized medicine are driving growth in the rare disease genetic testing market. The rising burden of cystic fibrosis, Duchenne muscular dystrophy, Huntington’s disease, lysosomal storage disorders, fragile X syndrome, and others inflates the demand for genetic testing in the global market. These tests help in early diagnosis, treatment planning, and therapeutic approaches for patients suffering from rare genetic disorders. In addition, government initiatives and funding for genetic research are further fueling the market demand globally.
According to the February 2024 WHO report, rare cancers with fewer than six cases per 100,000 people annually account for 25% of all cancer diagnoses. The report further stated that lack of data slows progress in understanding the diseases, resulting in poor outcomes, with a 5-year survival rate of around 49% compared to 63% for common cancers. IARC is working on projects such as lungNENomics to improve diagnosis and treatment. Thus, growing cases are concerning research associations, pushing them to invest in the market.
Key Rare Disease Genetic Testing Market Insights Summary:
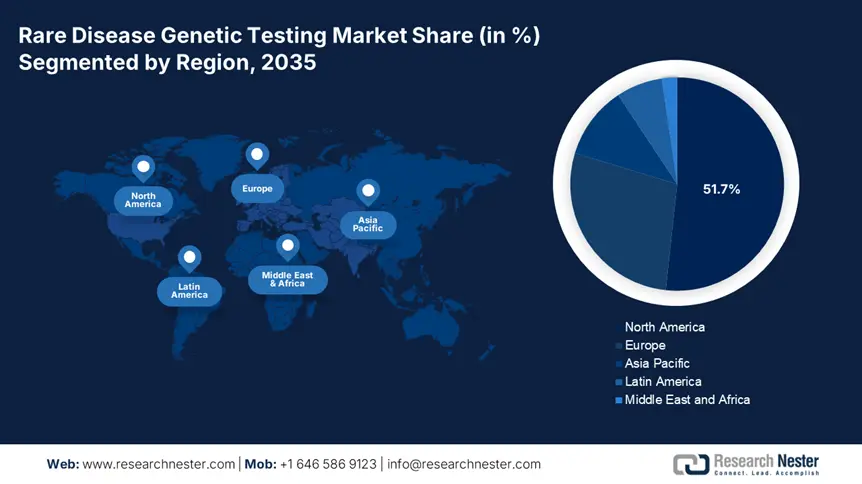
Regional Highlights:
- North America's 51.7% share in the Rare Disease Genetic Testing Market thrives due to advanced healthcare infrastructure and strong demand for personalized genetic testing services, ensuring growth through 2035.
- Asia Pacific's Rare Disease Genetic Testing Market is expected to see the fastest growth by 2035, attributed to rising burden of rare diseases and improvements in diagnostic services by regional pharmaceutical firms.
Segment Insights:
- The Endocrine & Metabolism Diseases segment is expected to capture a significant share from 2026-2035, propelled by the need for early and accurate diagnosis of genetically linked endocrine diseases.
- The Next-Generation Sequencing segment of the Rare Disease Genetic Testing Market is forecasted to capture over 39.8% share by 2035, propelled by its high-throughput and cost-effective capabilities for rare disease diagnosis.
Key Growth Trends:
- Growing government investments
- Technological advancements in genetic testing
Major Challenges:
- Limitations in affordability and accessibility
- Complexity of data interpretation
- Key Players: Centogene N. V, Arup Laboratorie, Eurofins Scientific, Strand Life Sciences, Ambry Genetics, Perkin Elmer, Inc., Realm IDX, Inc..
Global Rare Disease Genetic Testing Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 1.29 billion
- 2026 Market Size: USD 1.47 billion
- Projected Market Size: USD 5.36 billion by 2035
- Growth Forecasts: 15.3% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (51.7% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, United Kingdom, Japan, France
- Emerging Countries: China, India, Japan, South Korea, Brazil
Last updated on : 12 August, 2025
Rare Disease Genetic Testing Market Growth Drivers and Challenges:
Growth Drivers
- Growing government investments: The increased demand for early diagnosis and prevention is making genetic testing a preferable investment option for public and private organizations worldwide. For instance, in July 2022, the Ministry of Health and Family Welfare announced the launch of the National Policy for Rare Diseases (NPRD) 2021 to support rare disease diagnosis and treatment. Under this policy, eight Centers of Excellence have been set up, and financial aid up to INR 50 Lakhs (USD 58,283.5) is provided for treatment. Such initiatives improve access to disease care, encouraging the rare disease genetic testing to expand.
- Technological advancements in genetic testing: With advancements in genetic testing, the frequency of innovation in the rare disease genetic testing market increases. The substantial advancements in CRISPR gene editing and AI-driven diagnostics are enhancing accuracy and efficacy in the treatment of rare diseases. In May 2021, CENTOGENE N.V. reported that with the use of Bio/Databanks and deep genetic analysis, the company discovered 6 new rare diseases and 31 additional candidate genes. Thus, it reinforces the role of advanced technologies in driving market growth.
Challenges
- Limitations in affordability and accessibility: One of the biggest challenges in the rare disease genetic testing market is the high cost of advanced genetic tests, which can create an economic barrier among patients from price-sensitive regions. This may limit the expansion of this sector. In addition, tests like whole genome sequencing and whole exome sequencing can be expensive and may not be covered under insurance, restricting access to patients in underdeveloped countries. Moreover, the limited availability of genetic testing can delay the diagnosis of rare disorders, restraining market expansion.
- Complexity of data interpretation: Despite having vast amounts of complex data, genetic testing interprets a major challenge for healthcare providers due to which rare disease genetic testing market growth can be hindered. Incomplete genetic databases can lead to unclear or inconclusive results, delaying diagnosis and treatment. Additionally, the lack of global standardization in genetic testing protocols and regulatory frameworks can further create a hurdle to captivate the optimum consumer base in the genetic testing industry, limiting its widespread use for rare diseases.
Rare Disease Genetic Testing Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
CAGR |
15.3% |
|
Base Year Market Size (2025) |
USD 1.29 billion |
|
Forecast Year Market Size (2035) |
USD 5.36 billion |
|
Regional Scope |
|
Rare Disease Genetic Testing Market Segmentation:
Technology (Next-Generation Sequencing, Array Technology, PCR-Based Testing, FISH, Sanger Sequencing, Karyotyping)
Next-generation sequencing segment is predicted to capture over 39.8% rare disease genetic testing market share by 2035. The domination is majorly due to its ability to provide high-throughput and cost-effective genetic analysis. The segment is pledged with an increased need for identifying complex mutations associated with rare diseases, significantly improving diagnostic accuracy. For instance, in October 2021, CENTOGENE N.V. and Twist Bioscience Corporation partnered to develop advanced sequencing tools to make genetic testing more accessible for patients with rare diseases. Such collaborations highlight the increasing reliance on NGS-based solutions, ultimately driving market growth.
Disease type (Endocrine & Metabolism Diseases, Immunological Disorders, Neurological Diseases, Hematology Diseases, Cancer, Musculoskeletal Disorders, Cardiovascular Disease)
Based on disease type, the endocrine and metabolism diseases segment is projected to garner a significant share of the rare disease genetic testing market during the forecast period. This segment is dominated by the growing need for genetic testing for such diseases. The conditions often require early and precise genetic detection for effective management. In April 2024, according to a study by Annales d'Endocrinologie on multiple endocrine neoplasia (MEN), endocrine tumors are associated with genetic factors, making early diagnosis and treatment crucial, with genetic testing confirming 90% of diagnoses. The study underscores the demand for testing in the market to detect rare diseases.
Our in-depth analysis of the global rare disease genetic testing market includes the following segments:
|
Technology |
|
|
Disease Type |
|
|
Specialty |
|
|
End User |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Rare Disease Genetic Testing Market Regional Analysis:
North America Market Analysis
North America in rare disease genetic testing market is predicted to capture over 51.7% revenue share by 2035. Advanced healthcare infrastructure with the contribution of predictive and presymptomatic testing that is used for diagnosing gene mutations in rare diseases are major growth factors in this landscape. In January 2020, according to an article published in PNAS, approximately 2.7 billion people worldwide that is, 36% of the population were carriers of at least one mutation that can cause AR-IRD. Thus, the study highlights the growing need for predictive and presymptomatic testing, such as rare disease genetic testing, in this region.
The U.S. has become the hub for global leaders in the rare disease genetic testing market due to its wide consumer base and excellent demand for personalized genetic testing services. The country offers a great opportunity for companies to expand their business, inspiring other domestic participants to establish their footprint in this country by promoting the effectiveness of this testing. For instance, in June 2021, Eurofins Scientific announced its acquisition of DNA Diagnostics Center to enhance the capabilities of genetic testing and expand its consumer base in the region. The acquisition further underscores the market expansion during the forecast timeline.
Canada is steadily consolidating its position in the rare disease genetic testing market with proactive requirements of accurate detection and technological advancements. The country’s genetic testing industry is further driven by collaborations between healthcare facilities and diagnostic services. In August 2024, Quest Diagnostics announced the acquisition of LifeLabs, enhancing the accessibility to diagnostic innovation, and the deal was valued at approximately USD 1 billion. Therefore, this is further inflating demand in this sector due to the proven demand for genetic testing.
APAC Market Statistics
Asia Pacific is anticipated to demonstrate the fastest growth in the rare disease genetic testing market with its presence of countries with underdeveloped areas. The region has a rising burden of rare diseases, showcasing substantial market expansion. Companies delivering genetic testing services are both accessible and affordable, raising their demand and offering a broader consumer base in this sector. Thus, the growth in this region is further carried forward with the efforts of regional pharmaceutical firms that are making improvements in the diagnosis sector.
India is propagating the regional rare disease genetic testing market with its technological advancements, awareness of personalized medicine, and the burden of increasing genetic disorders. For instance, in February 2025, MedGenome launched the #CarefortheRare campaign in the country to promote early genetic testing for rare diseases. The campaign further highlighted the role of genomic diagnostics in identifying inherited conditions. Such initiatives reflect the country’s focus on rare disease management with early detection.
China has one of the biggest rare disease genetic testing market due to expansion in healthcare investments, genomic trends and the largest patient pool. The country’s government and private sector initiatives raise awareness about the diseases to improve testing efficiency. In June 2024, Illumina launched the iHope China program to provide genetic testing for 1800 families affected by rare diseases. Such programs expand access to genetic diagnostics across the country, further highlighting the requirement.
Key Rare Disease Genetic Testing Market Players:
- Quest Diagnostics Inc.
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent developments
- Regional Presence
- SWOT Analysis
- Invitae Corp
- Eurofins Scientific
- Centogene N. V
- Arup Laboratories
- Eurofins Scientific
- Strand Life Sciences
- Ambry Genetics
- Perkin Elmer, Inc.
- Realm IDX, Inc.
- Macrogen, Inc.
- Baylor Genetics
- Color Genomics, Inc
- Biogen Inc.
The competitive demographic of the rare disease genetic testing market is inspiring global leaders to strengthen their technological and research capabilities. With continuous investments in diagnostic tools and genomic innovations, companies are expanding their global reach. In March 2023, Invitae announced a partnership with Epic to enhance genetic testing workflows for rare disorders through Epic’s Aura diagnostic suite test results are efficiently integrated into the clinical systems. With a keen eye for detail on testing accuracy, reducing turnaround times, and further widening the market’s scope during the forecast period.
Below is the list of some key players in the market:
Recent Developments
- In May 2024, Biogen Inc. received European Commission approval for QALSODY® (tofersen), the first therapy for SOD1-ALS, a rare genetic form of amyotrophic lateral sclerosis (ALS) that affects approximately 1,000 people in Europe.
- In April 2023, 3billion announced that it received accreditation from the CDPH as a clinical laboratory in the U.S. for genetic testing. The accreditation also allows insurance claims for genetic tests, improving accessibility.
- In January 2023, Premier Research and CENTOGENE N.V. announced their partnership to accelerate clinical trials for rare diseases, reducing risks and streamlining the development process.
- Report ID: 7419
- Published Date: Aug 12, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Rare Disease Genetic Testing Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




