Personalized Cell Therapy Market Outlook:
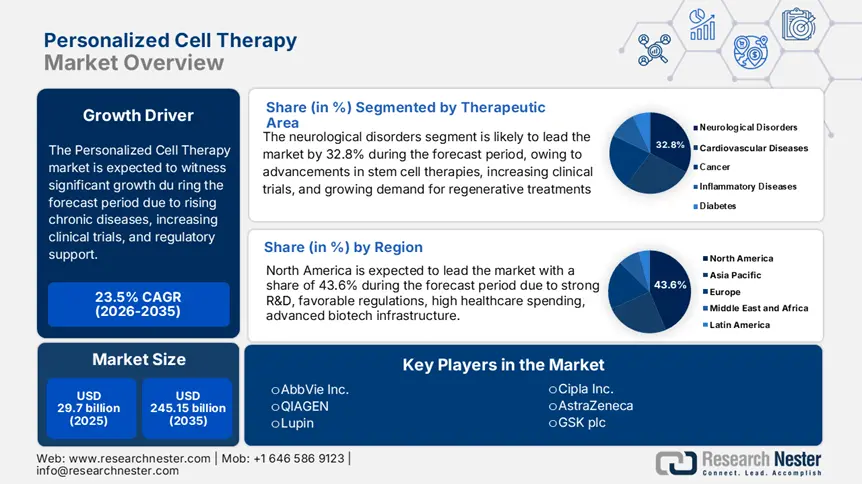
Personalized Cell Therapy Market size was over USD 29.7 billion in 2025 and is projected to reach USD 245.15 billion by 2035, witnessing around 23.5% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of personalized cell therapy is evaluated at USD 35.98 billion.
The rising prevalence of cancer, autoimmune diseases, and neurological disorders is significantly driving the growth of the personalized cell therapy market. As per NLM in February 2024, estimates of the yearly increases in the overall worldwide incidence and prevalence of autoimmune diseases are 19.1% and 12.5%, respectively. Traditional treatments often have limited effectiveness or severe side effects, whereas personalized cell therapies offer targeted, patient-specific solutions that enhance efficacy and minimize adverse reactions. With the increasing global disease burden, there is a growing demand for innovative therapies that provide long-term benefits. This demand fuels research, investment, and commercialization, accelerating market expansion and adoption.
Additionally, innovations such as CRISPR, CAR-T cell therapy, and stem cell modifications are revolutionizing personalized cell therapy by enhancing treatment precision, improving patient outcomes, and expanding therapeutic applications. These advancements enable targeted gene editing, boosting the effectiveness of therapies for cancer, genetic disorders, and autoimmune diseases. As these technologies become more refined and widely adopted, they drive investment, regulatory approvals, and commercialization, accelerating the growth and accessibility of the personalized cell therapy market globally.
Key Personalized Cell Therapy Market Insights Summary:
Regional Highlights:
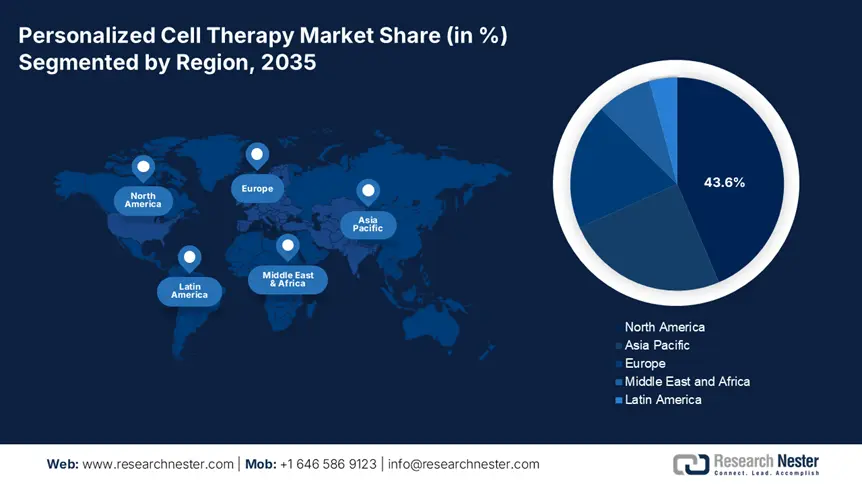
- North America dominates the Personalized Cell Therapy Market with a 43.6% share, driven by the region’s strong biotech ecosystem and a high incidence of cancer, solidifying its leading position through 2035.
- APAC's personalized cell therapy market is projected to achieve the fastest CAGR by 2035, driven by multinational pharma and biotech firms investing in the region and rapid healthcare advancements.
Segment Insights:
- The Neurological Disorders segment is forecasted to capture 32.8% market share by 2035, propelled by the increasing prevalence of neurodegenerative diseases and the shift toward precision medicine for neural tissue repair and improved patient outcomes.
- Lymphocytes (Cell Type) segment are projected to hold the majority share by 2035, driven by the increasing adoption of CAR-T cell therapies for cancer treatment and advancements in immunotherapy.
Key Growth Trends:
- Growing demand for regenerative medicine
- Expanding healthcare infrastructure
Major Challenges:
- Volatility and disparity in product pricing
- Limitation in hiring adequate workforce
Key Players: Cipla Inc., Lupin, Sun Pharmaceuticals Industries Ltd., Hikma Pharmaceuticals PLC.
Global Personalized Cell Therapy Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 29.7 billion
- 2026 Market Size: USD 35.98 billion
- Projected Market Size: USD 245.15 billion by 2035
- Growth Forecasts: 23.5% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (43.6% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Japan, United Kingdom, France
- Emerging Countries: China, India, Japan, South Korea, Singapore
Last updated on : 12 August, 2025
Personalized Cell Therapy Market Growth Drivers and Challenges:
Growth Drivers
- Growing demand for regenerative medicine: The growing emphasis on regenerative medicine for repairing damaged tissues and organs is driving demand for personalized cell therapies. Stem cell-based treatments are increasingly used to regenerate tissues affected by conditions such as neurodegenerative disorders, cardiovascular diseases, and musculoskeletal injuries. These therapies promote tissue repair, reduce inflammation, and enhance functional recovery. As regenerative medicine gains traction for its potential to provide long-term solutions, the adoption of personalized cell therapies continues to expand, fueling market growth.
- Expanding healthcare infrastructure: The expansion of healthcare infrastructure and increased access to advanced therapies in emerging markets are driving the growth of personalized cell therapy. With the development of specialized cell therapy centers and expanding hospital networks, more patients can access innovative treatments. Improved diagnostic capabilities and better clinical facilities also enhance the effectiveness and availability of cell-based therapies. This increased accessibility is fostering wider adoption, making personalized cell therapy more viable and contributing to the expansion of the personalized cell therapy market.
Challenges
- Volatility and disparity in product pricing: The personalized nature of autologous (patient-specific) cell therapies often leads to high manufacturing costs, making them financially burdensome for patients and healthcare systems. These therapies require extensive processes such as cell collection, manipulation, and reinfusion, which contribute to the overall expenses. As a result, while these treatments can offer tailored solutions for patients, the financial implications pose significant challenges in accessing and providing such advanced medical options on a wider scale.
- Limitation in hiring adequate workforce: The ongoing shortage of skilled workers in crucial sectors such as manufacturing, analytical development, testing, and quality control poses significant challenges for the cell therapy industry. This gap in expertise not only affects the efficiency of development processes but also delays the production of vital therapies. As these skilled roles are essential for ensuring rigorous quality standards and effective testing protocols, the lack of personnel can hinder advancements in cell therapies, ultimately impacting patient access to innovative treatments.
Personalized Cell Therapy Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
CAGR |
23.5% |
|
Base Year Market Size (2025) |
USD 29.7 billion |
|
Forecast Year Market Size (2035) |
USD 245.15 billion |
|
Regional Scope |
|
Personalized Cell Therapy Market Segmentation:
Therapeutic Area (Neurological Disorders, Cardiovascular Diseases, Inflammatory Diseases, Diabetes, Cancer)
Based on therapeutic area, the neurological disorders segment is expected to account for more than 32.8% personalized cell therapy market share by the end of 2035. The segment’s growth is attributed to the increasing prevalence of neurogenerative diseases. The number of people affected from nervous system ailments around the globe was 3.4 billion in 2021: Institute for Health Metrics and Evaluation. Personalized therapies offer potential for repairing damaged neural tissues, improving patient outcomes, and addressing unmet medical needs, making them a first-line treatment for this segment. Additionally, the shift toward precision medicine and patient-specific treatments further drives adoption in neurological disorder applications, fueling the market.
Cell Type (Lymphocytes, Mesenchymal Stem Cell, Hematopoietic Stem Cell, Others)
By cell type segment, the lymphocytes segment is slated to garner the majority personalized cell therapy market share over the forecast period. The segment is growing due to the rising adoption of CAR-T cell therapies for cancer treatment. Advancements in immunotherapy, increasing cancer prevalence, and regulatory approvals for lymphocyte-based therapies drive demand. Additionally, ongoing clinical trials, expanding research in autoimmune diseases, and improved genetic engineering techniques contribute to growth. The shift toward targeted, patient-specific treatments further accelerates the adoption of lymphocyte-based personalized cell therapies, enhancing efficiency and patient outcomes.
Our in-depth analysis of the global market includes the following segments:
|
Therapeutic Area |
|
|
Cell Type |
|
|
End-users
|
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Personalized Cell Therapy Market Regional Analysis:
North America Market Statistics
North America in personalized cell therapy market is expected to account for around 43.6% revenue share by 2035. The region has a high incidence of cancer. The cancer incidence in the U.S. is projected to increase, with an estimated 2 million new cases in 2025: American Cancer Society. Autoimmune diseases and genetic disorders are also driving the demand for targeted, patient-specific treatments for improved outcomes over traditional methods. The region’s strong biotech ecosystem, including leading research institutions and universities, fosters continuous innovation in CAR-T cell therapy, stem cell applications, and gene editing. These advancements enhance treatment precision, expand therapeutic applications, and attract investment.
The U.S. leads the personalized cell therapy market with its strong emphasis on clinical trials. With increasing investments in R&D, innovations in new applications, attaining successful trials, lead to accelerated regulatory approvals, propelling market growth. Additionally, advancements in cell culture, bioprocessing automation, and scalable manufacturing techniques are reducing production costs, making therapies more accessible. Improved logistics for cell-based treatments further enhance distribution and availability, supporting broader commercialization and expanding the personalized cell therapy market across various medical applications.
The aging population in Canada is increasingly affected by chronic and degenerative diseases, driving demand for advanced, long-term treatment solutions such as personalized cell therapies. As per the 2020 report from the government of Canada, 73% of individuals aged 65+ years had at least 1 of 10 common chronic diseases. Simultaneously, growing awareness and acceptance of regenerative medicine are accelerating the adoption of cell-based immunotherapies for cancer, neurological disorders, and rare genetic diseases. This rising demand encourages investment in research, regulatory approvals, and commercialization efforts, expanding the personalized cell therapy market and positioning Canada as a key player in regenerative medicine advancements.
APAC Market Analysis
The APAC region is expected to garner the fastest CAGR over the forecast period. Multinational pharma and biotech firms are actively investing in the Asia Pacific personalized cell therapy market. They are fostering strategic partnerships for technology transfers and commercial escalation. Simultaneously, rapid healthcare advancements, improved medical infrastructure, and rising healthcare spending are enhancing accessibility to these advances. This combination of foreign investments and healthcare improvements is driving innovation, expanding treatment availability, and increasing patient adoption, ultimately fueling the growth of the personalized cell therapy market across the region.
Significant investments in healthcare infrastructure and advanced medical technologies in China are enhancing accessibility to personalized cell therapies, allowing a larger patient population to benefit from innovative treatments. Government expenditure on healthcare services in China increased from 49.6% in 2019 to 58.2% in 2022: Frontiers. Additionally, the National Medical Products Administration (NMPA) has streamlined approval processes, expediting clinical trials and commercialization of cell-based therapies. These combined efforts are driving faster innovation, increasing patient adoption, and encouraging biotech companies to expand their research and production, ultimately fueling the growth of the personalized cell therapy market in China.
The government and private investors in India are heavily funding biotech research, driving advancements in stem cell therapy, CAR-T cell therapy, and gene editing technologies. India's biotechnology sector is crucial for economic growth, with an estimated contribution of USD 130 billion in 2024, which is projected to reach USD 300 billion by 2030: IBEF. Simultaneously, rapid improvements in hospitals, research centers, and medical infrastructure are enhancing accessibility to personalized cell therapies across urban and semi-urban areas. This combination of financial support and healthcare expansion is accelerating innovation, increasing patient adoption, and strengthening the position of India in the growing personalized cell therapy market.
Key Personalized Cell Therapy Market Players:
- QIAGEN
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- AbbVie Inc.
- Bausch Health Companies Inc.
- Teva Pharmaceutical Industries Ltd.
- Cipla Inc.
- Lupin
- Sun Pharmaceuticals Industries Ltd.
- Hikma Pharmaceuticals PLC
- AstraZeneca
- GSK Plc.
- Pfizer Inc.
- Amneal Pharmaceuticals LLC.
Key companies in the personalized cell therapy market are driving innovation through advancements in CAR-T cell therapy, stem cell research, and gene editing technologies such as CRISPR. They are improving bioprocessing automation, scaling up manufacturing, and reducing costs to enhance accessibility. The collaborative effort from biotech firms, research institutions, and regulatory bodies is accelerating clinical trials and approvals. Additionally, investments in AI-driven cell analysis and personalized treatment algorithms are optimizing patient-specific therapies, fueling market growth and adoption. Such key players are:
Recent Developments
- In March 2025, AstraZeneca was set to acquire EsoBiotec, a biotech firm developing the ENaBL platform, which enables rapid, in vivo cell therapies to empower immune responses against cancer.
- In January 2025, Bristol Myers Squibb Canada announced Health Canada’s approval of Breyanzi (lisocabtagene maraleucel) for adult patients with refractory DLBCL, PMBCL, HGBCL, or DLBCL from follicular lymphoma, eligible for HSCT.
- Report ID: 7443
- Published Date: Aug 12, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Personalized Cell Therapy Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




