Immune Thrombocytopenia Market Outlook:
Immune Thrombocytopenia Market size was valued at USD 3.57 billion in 2025 and is set to exceed USD 4.53 billion by 2035, expanding at over 2.4% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of immune thrombocytopenia is estimated at USD 3.65 billion.

The impact of immune thrombocytopenia (ITP) on children and adolescents, particularly who are aged between 2 and 5, around the globe is a serious health concern for authorities. The fast progression and severity of this condition are propelling demand for efficient curative and management therapeutics. As per an NLM article from May 2024, 1-6.4 children in every 100,000 population in the world were diagnosed with this autoimmune disorder. It further highlighted its chronic characteristics among people aged 60, accounting for a prevalence of 12 per 100,000. Further, annual increments in cases are expanding the patient pool, securing a continuous surge in the immune thrombocytopenia (ITP) market.
The asymptomatic hospitalization stage of this condition adds up to the total healthcare expenditure on affected individuals. On this note, an observational cost analysis from NLM, published in July 2024, revealed that the total spending on primary and chronic ITP (cITP) each year in Europe was USD 11621.2 and USD 21432.4, respectively. The classification of mean annual cost further testified higher economic burden on chronically ill residents. Thus, the recent R&D cohorts in the ITP market are focusing on the standardization of more cost-effective payers’ pricing for medicines. In addition, their intentions on making early diagnosis a regular practice in every medical setting is also promoting wide adoption by increasing accessibility.
Distribution of Mean Annual Cost of cITP in Europe (2020)
|
Area of Expenditure |
Mean Annual Cost (in USD) per Patient |
|
Medication |
12547.4 |
|
Hospitalization |
5588.2 |
|
Specialized Care |
964.0 |
|
Primary Care |
386.2 |
|
Total |
21432.4 |
Source: NLM Study
Key Immune Thrombocytopenia (ITP) Market Insights Summary:
Regional Highlights:
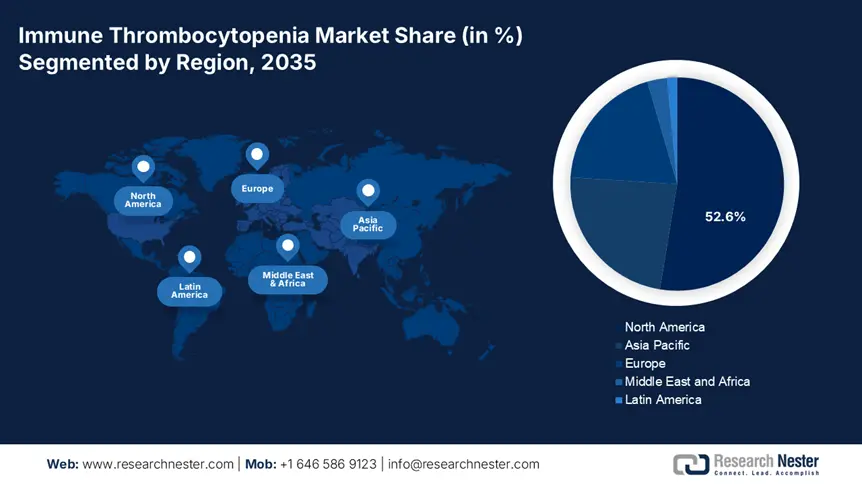
- North America commands a 52.6% share in the Immune Thrombocytopenia Market, fueled by a well-established healthcare infrastructure and strong emphasis on clinical discoveries, ensuring growth through 2035.
- Asia Pacific’s immune thrombocytopenia market is poised for rapid growth by 2035, driven by recent healthcare developments and pharmaceutical expansion in developing economies.
Segment Insights:
- Hospitals and Clinics segment are anticipated to hold a dominant share by 2035, fueled by the need for hospitalization in managing immune thrombocytopenia.
- Acute Immune Thrombocytopenia segment is projected to hold a 68.5% share by 2035, driven by higher occurrence in children and effective novel product pipelines.
Key Growth Trends:
- Ongoing discoveries of innovative therapeutics
- Increasing focus on clinical trials and compliance
Major Challenges:
- Lack of awareness about the available options
- High fragmentation and competition in the market
- Key Players: Pfizer Inc., Novartis AG, Sanofi, Janssen Pharmaceuticals Inc., Amgen Inc., Jiangsu Hengrui Pharmaceutical Co. Ltd., Teva Pharmaceutical Industries Ltd.Hoffmann-La Roche Ltd.
Global Immune Thrombocytopenia (ITP) Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 3.57 billion
- 2026 Market Size: USD 3.65 billion
- Projected Market Size: USD 4.53 billion by 2035
- Growth Forecasts: 2.4% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (52.6% Share by 2035)
- Fastest Growing Region: Europe
- Dominating Countries: United States, Germany, Japan, United Kingdom, France
- Emerging Countries: China, India, Japan, South Korea, Brazil
Last updated on : 12 August, 2025
Immune Thrombocytopenia Market Growth Drivers and Challenges:
Growth Drivers
- Ongoing discoveries of innovative therapeutics: The progression in the immune thrombocytopenia market is highly driven by continuous clinical findings. The introduction of more specific detection methods and effective therapies, such as biologics, small molecules, and agonists, are improving patient outcomes. This personalized approach is fostering diversity and new scopes of business. For instance, in March 2024, Sobi unleashed the potential of avatrombopag (Doptelet) in treating pediatric ITP cases, with proven safety and efficacy from the AVA-PED-301 study. The analyzed 6-month episode in this trial concluded that this oral treatment outperformed placebo with 28.0% greater durability in platelet response.
- Increasing focus on clinical trials and compliance: As more pharma companies put their effort into globalization, the immune thrombocytopenia (ITP) market tends to generate more revenue. They are utilizing advances in clinical trials and drug formulations to attain maximum acceptance across emerging healthcare territories, such as Japan, Canada, and India. This can be testified by the upscaling sales of Tavalisse (fostamatinib disodium hexahydrate) after earning regulatory allowances from Japan (2022) and Canada (2020). In 2022, Rigel Pharmaceuticals recorded the highest quarterly shipment of 2,417 bottles in the U.S., marking a net sales growth of USD 4.3 million from 2021. This trend is further followed by others to support market expansion.
Challenges
- Lack of awareness about the available options: Despite the positive impact of current promotional campaigns, this condition still remains under-recognized due to being relatively rare. Particularly, in resource-constrained regions, such as Africa, delayed diagnosis and suboptimal management are severely impacting the global widespread of the immune thrombocytopenia market. Furthermore, the limitations in understanding this disease and in local financial support, resulting from the lack of proper knowledge, are restricting the sector’s growth.
- High fragmentation and competition in the market: Variability in medical infrastructures and reimbursement policies across different regions may foster an economic and availability hurdle in the ITP market. In addition, underserved areas often lag in accommodating adequate facilities for complete diagnosis and treatment, limiting distribution channels. Furthermore, the highly competitive scenario of this field may also encompass trading difficulties for new entrants, restraining engagement from both consumers and participants.
Immune Thrombocytopenia Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
CAGR |
2.4% |
|
Base Year Market Size (2025) |
USD 3.57 billion |
|
Forecast Year Market Size (2035) |
USD 4.53 billion |
|
Regional Scope |
|
Immune Thrombocytopenia Market Segmentation:
Type (Acute Immune Thrombocytopenia, Chronic Immune Thrombocytopenia)
Based on type, the acute immune thrombocytopenia segment is estimated to capture over 68.5% immune thrombocytopenia market share by 2035. This subtype has a higher occurrence in children and better medicinal recovery rate, making it the major contributor to this sector’s enlargement. The novel product pipelines of the immune thrombocytopenia market are notably effective for the associated patients, placing this segment as the top priority for global leaders. Worldwide awareness initiatives and funding for saving the younger generation from such autoimmune disorders are also fueling this segment’s leadership. Furthermore, the fewer adverse events in treating this type of ITP helps accelerate regulatory approvals, promoting wide use and easy availability of related medicines.
End users (Hospitals and Clinics, Specialty Centers, Research and Academic Institutes)
In terms of end users, the hospitals and clinics segment is predicted to dominate the immune thrombocytopenia (ITP) market over the analyzed period. The unavoidable need for hospitalization in managing this immune-mediated bleeding disease pushes this segment to generate the maximum revenue. The NLM article from July 2023 observed an annual increment in hospital admissions for immune thrombocytopenia across the U.S., particularly for adults. This segment’s proprietorship can be testified by the emphasizing economic contribution of these organizations in emerging healthcare marketplaces. For instance, in 2023, IBEF reported that the industry value of hospitals in India accounted for USD 98.9 billion, exhibiting a CAGR of 8.0%. It was also estimated to be valued at USD 193.5 billion by 2032.
Our in-depth analysis of the global immune thrombocytopenia market includes the following segments:
|
Type |
|
|
Treatment |
|
|
End users |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Immune Thrombocytopenia Market Regional Analysis:
North America Market Analysis
North America immune thrombocytopenia market is set to hold revenue share of more than 52.6% by 2035. The region’s leadership is driven by two major factors, such as a well-established healthcare infrastructure and a strong emphasis on clinical discoveries. In search of advanced, targeted, and tailored treatment options, many are conducting rigorous R&D studies. For instance, in December 2024, Seismic Therapeutic displayed promising preclinical data on S-1117 at the annual meeting of the American Society of Hematology (ASH). This pan-immunoglobulin G (IgG) sculpting enzyme candidate surpassed the level of efficacy of neonatal Fc receptor (FcRN) inhibitor therapy in treating acute ITP.
The U.S. ITP market is pledged with the presence of internationally recognized research-based pharma pioneers who are cultivating an in-house innovation culture. Besides, the country consists of a large consumer base with a heavy economic burden. On this note, an NLM study from February 2020 concluded that the rate of annual ITP incidence in the U.S. is 6.1 per 100,000, where around 20,000 new cases get registered every year. It also mentioned the overall spending of these patients in this country to be USD 400.0 million. This is pushing both public and private organizations to increase accessibility in this sector.
Canada is propelling augmentation in the immune thrombocytopenia market with favorable government initiatives and subsidiary policies. In the desperate efforts to attain the highest quality in healthcare services, the governing bodies of this country are heavily investing in this medical discipline. For instance, in March 2023, the Government of Canada allocated USD 1.5 billion over 3 years for the National Strategy for Drugs for Rare Disease. Then again, in February and March 2025, USD 48.0 million and USD 10.0 million were designated to the habitats of Manitoba and Prince Edward Island (PEI), respectively, serving the same purpose. Such events are securing a steady capital influx in this sector.
APAC Market Statistics
Asia Pacific is poised to register the fastest growth in the immune thrombocytopenia market over the forecasted period. Concerning demographics, recent healthcare developments, and pharmaceutical expansion, these are conjugatively propelling its pace of progression. Developing economies such as Japan, China, and India are meticulously working on the improvement of public access to advanced medical services. This is inspiring global pharma producers to penetrate their offerings with precise diagnosis and long-acting regimens. For instance, in January 2025, CASI Pharmaceuticals announced the commencement of patient dosing in the phase 1/2 trial on CID-103. The evaluation was intended to obtain regulatory clearance in China for adults with cITP.
India is propagating in the ITP market with government support in domestic drug production and biotechnology expansion. The country presents its global leadership in R&D with excellence in the life sciences industry. Besides the initiatives for local advances in innovation and supply chain, its nationwide frameworks are also open to international contributions. According to the Investment Grid of India, the nation’s medical infrastructure industry possessed over 600 opportunities for investment in 2021, totaling around USD 32.0 billion. Furthermore, the recently introduced data-driven methodology is escalating the nation’s R&D participation in this sector.
China is a lucrative landscape for global pioneers in the immune thrombocytopenia market, presenting a large consumer base and a great manufacturing capacity. In addition, the country utilizes its expertise in clinical trials and precision medicine to create a new line of products for this field. For instance, in January 2024, HUTCHMED successfully acquired priority review acceptance for a New Drug Application (NDA) for sovleplenib. The company attained support from the positive results from its previously conducted phase III ESLIM-01 trial in adult candidates with primary ITP, having previous therapy. This indicates the presence of a progressive business atmosphere in China.
Key Immune Thrombocytopenia Market Players:
- Pfizer Inc.
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Hoffmann-La Roche Ltd.
- Mylan N.V.
- Fresenius Kabi AG
- Hepalink Group.
- Teva Pharmaceutical Industries Ltd.
- Amarillo Biosciences Inc.
- Bolder Bio Technology Inc.
- GENOSCO Inc.
- Hansa Biopharma
- Janssen Pharmaceuticals Inc.
- CSL Limited
- Amgen Inc.
- Rigel Pharmaceuticals Inc.
- Dova Pharmaceuticals
- Novartis AG
- Sanofi
- Jiangsu Hengrui Pharmaceutical Co. Ltd.
- Shire and Ligand Pharmaceuticals Inc.
- Keymed Biosciences
The highly competitive dynamics of the immune thrombocytopenia market are influenced by ongoing discoveries. Key players in this sector are rigorously performing clinical studies to establish their products overseas. For instance, in September 2020, UCB unraveled the outcomes from the phase II study on rozanolixizumab, demonstrating remarkable improvements in the platelet count of residents with primary immune thrombocytopenia. The results showcased excellent adherence to patients, establishing a strong foundation for subcutaneously infused monoclonal FcRn antibody-based therapeutics in this field. Such innovations are encouraging other biopharma pioneers to bring more effective therapies with better safety profiles and reduced risk of recurrence. These key players are:
Recent Developments
- In January 2025, Keymed Biosciences launched a joint venture, Timberlyne Therapeutics, with Mountainfield Venture Partners. The new subsidiary was backed by a USD 180.0 million series A funding and positive phase I and II results for CM313 in treating refractory immune thrombocytopenia.
- In December 2024, Sanofi revealed promising benefits observed in the LUNA phase 3 study on rilzabrutinib, showcasing rapid and durable platelet response. This orally administered Bruton’s tyrosine kinase (BTK) inhibitor has the potential to cure persistent or chronic ITP, empowering its chances for regulatory approvals in the U.S. and Europe.
- Report ID: 7455
- Published Date: Aug 12, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




