Clinical Trial Investigative Site Network Market Outlook:
Clinical Trial Investigative Site Network Market size was valued at USD 9.27 billion in 2025 and is expected to reach USD 17.73 billion by 2035, expanding at around 6.7% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of clinical trial investigative site network is evaluated at USD 9.83 billion.

The clinical trial investigative site network market is expanding rapidly due to the synergy between drivers that amplify the need for efficient and well-executed clinical trials. For instance, in February 2025, in order to assess the effectiveness of THIO given in conjunction with a checkpoint inhibitor (CPI) in third-line non-small cell lung cancer (NSCLC) patients who are resistant to checkpoint inhibitors and chemotherapy, MAIA Biotechnology, Inc. announced plans to launch a Phase 3 pivotal trial in 2025 called THIO-104. Moreover, the rising complexity in current clinical trials, requires intensely specialized site networks that can successfully handle complex logistics and operational complexities.
At the same time, the increasing prevalence of chronic disorders across the world, such as oncology, cardiovascular, and neurological conditions, accelerates demand for new therapy interventions, adding to the size of clinical trials and the underlying need for durable site networks. For instance, in December 2024, the WHO revealed that 19 million deaths from NCDs in 2021 were attributable to cardiovascular illnesses, followed by cancer with 10 million cases, further adding to the cause, other chronic respiratory conditions with 4 million, and diabetes with over 2 million deaths. Thus, the market growth is orchestrated by the ubiquitous drive for the acceleration of drug development timelines.
Key Clinical Trial Investigative Site Network Market Insights Summary:
Regional Highlights:
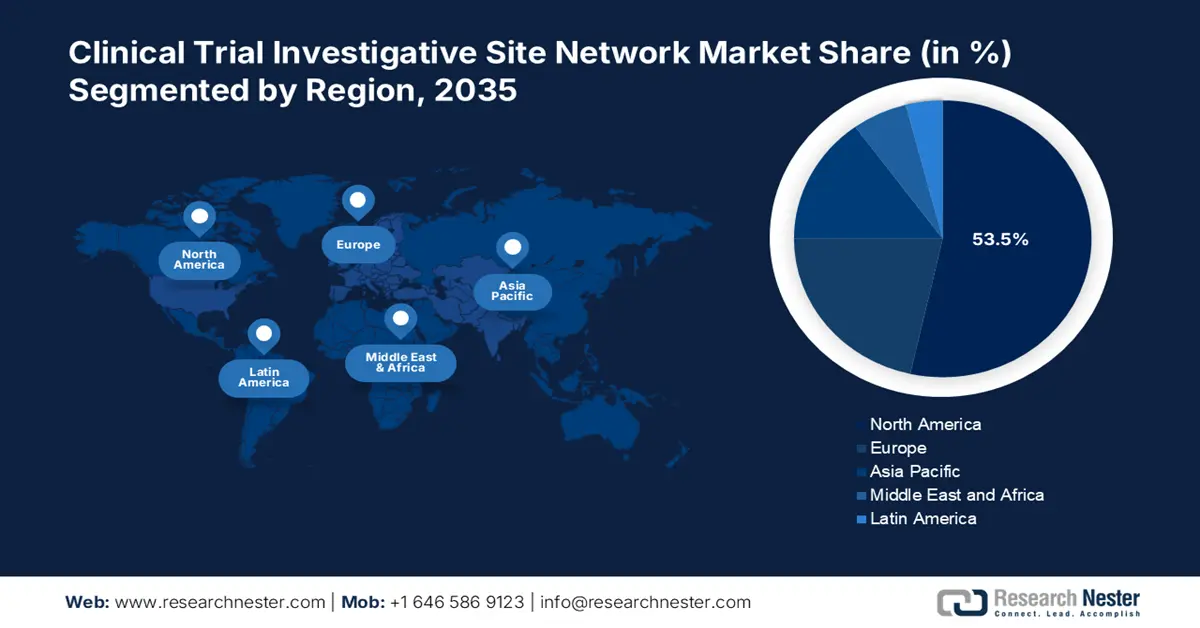
- North America leads with a 53.5% share in the Clinical Trial Investigative Site Network Market, driven by expanding portfolios and collaborations in biopharmaceutical sector, bolstering growth through 2035.
- Asia Pacific’s clinical trial investigative site network market is expected to expand exponentially by 2035, driven by ongoing development in clinical research and outsourcing companies.
Segment Insights:
- The Oncology segment is projected to capture a 36.6% share by 2035, fueled by the need for specialized expertise in complex, high-speed cancer trials.
- The Phase III segment of the Clinical Trial Investigative Site Network Market is projected to dominate the market from 2026 to 2035, fueled by large-scale confirmatory trials requiring robust site networks and protocol adherence.
Key Growth Trends:
- Decentralized and hybrid trial models
- Incorporation of AI & ML
Major Challenges:
- Site variability and quality control
- Cost pressure and resource constraints
- Key Players: Meridian Clinical Research, IQVIA Inc., Clinedge, WCG, ClinChoice, KV Clinicaland more.
Global Clinical Trial Investigative Site Network Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 9.27 billion
- 2026 Market Size: USD 9.83 billion
- Projected Market Size: USD 17.73 billion by 2035
- Growth Forecasts: 6.7% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (53.5% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, United Kingdom, Japan, Canada
- Emerging Countries: China, India, Japan, South Korea, Brazil
Last updated on : 12 August, 2025
Clinical Trial Investigative Site Network Market Growth Drivers and Challenges:
Growth Drivers
- Decentralized and hybrid trial models: The growth of the clinical trial investigative site network market is driven by essentially opening up patient accessibility. Furthermore, the elimination of geographic barriers with remote monitoring, telemedicine, and direct-to-patient services is fostering the growth. For instance, in March 2022, Kayentis announced the release of a fully integrated patient-centric platform that streamlines hybrid and DCT management. While significantly enhancing the way patients and sites use DCT technology, this simplified platform removes numerous logistical challenges in delivering essential clinical trial components to patients' homes.
- Incorporation of AI & ML: Through the optimisation of patient stratification and predictive analytics, the integration of artificial intelligence (AI) and machine learning (ML) in the clinical trial investigative site network market serves as a powerful growth driver. For instance, in January 2025, Lantern Pharma Inc. revealed developments in the use of its RADR AI platform to optimize and speed up the creation of antibody-drug conjugates (ADCs). This capability allows for the precise identification of ideal patient cohorts, greatly improving recruitment efficiency and cutting down on trial timelines. Consequently, the delivery of clinically relevant data speeds up and promotes more focused therapeutic development.
Challenges
- Site variability and quality control: The inherent variability of data gathering activities, employees' expertise, and working protocols in geographically isolated investigative locations is a priority concern in the clinical trial investigative site network market. This unreliability later destroys the integrity of clinical trial results by way of patient care deviations, data precision at entry points, and following a protocol. This issue is mostly a consequence of the decentralized nature of these networks, differences in site infrastructures, training levels, and resource allocation lead to dramatic differences in trial conduct. Hence, these are compounded by the lack of standard operating procedures and centralized monitoring systems and threaten the validity of study findings.
- Cost pressure and resource constraints: The clinical trial investigative site network market is greatly hindered by the increasing costs of more complex trial protocols and technology integrations. The primary reason for this challenge is the increasing demand for specialized personnel and advanced technologies, including advanced data management systems and remote monitoring tools, which entail huge financial outlays. These increasing expenses, coupled with the reality that numerous investigative sites have budgetary restrictions, result in resource shortages that ensure it becomes challenging to conduct clinical trials optimally. This may delay drug development timelines and render it increasingly hard to sustain high standards for patient management and data collection.
Clinical Trial Investigative Site Network Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
CAGR |
6.7% |
|
Base Year Market Size (2025) |
USD 9.27 billion |
|
Forecast Year Market Size (2035) |
USD 17.73 billion |
|
Regional Scope |
|
Clinical Trial Investigative Site Network Market Segmentation:
Therapeutic Areas (Oncology, CNS, Pain Management, Endocrine, Cardiology)
By 2035, oncology segment is estimated to capture clinical trial investigative site network market share of over 36.6%, driven by the need for cancer studies to keep up with the speed, combined with the complexity of cancer trials needing expertise in the site. For instance, in March 2025, to promote cooperation among clinical trial sponsors, study sites, contract research organizations (CROs), and patient advocates, Advarra announced the launch of Oncology Research Pulse. It is a new content-driven, thought leadership initiative to facilitate a forum for knowledgeable opinions, insightful discussions, and the sharing of best practices.
Phase (Phase I, Phase II, Phase III, Phase IV)
The phase III segment is anticipated to dominate the clinical trial investigative site network market by 2035 attributable primarily to the massive number of confirmatory, large-scale trials that are needed for regulatory approval and marketplace access. For instance, in January 2024, -Merck declared that four of its investigational compounds from its broad pipeline in solid tumors and hematologic malignancies had begun pivotal Phase 3 trials. The following investigational candidates are now being enrolled in global Phase 3 studies: Bomedemstat, Nemtabrutinib, MK-2870, and MK-5684. The large patient population trials involving complicated logistical requirements call for robust site networks capable of supporting voluminous data gathering and strict protocol adherence fueling demand for specialized site management skills.
Our in-depth analysis of the global clinical trial investigative site network market includes the following segments:
|
Therapeutic Areas |
|
|
Phase |
|
|
End use |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Clinical Trial Investigative Site Network Market Regional Analysis:
North America Market Statistics
North America in clinical trial investigative site network market is predicted to account for more than 53.5% revenue share by the end of 2035, attributable to the expanding portfolios of present companies in the region. For instance, in July 2024, to serve the US biopharmaceutical business, SGS North America announced the expansion of its biologics testing services, increasing its capacity and capabilities. This advancement seeks to expedite the delivery of biologic products and provide clients with enhanced testing options.
The U.S. clinical trial investigative site network market is likely to unfold remarkable growth opportunities during the projected timeline owing to the collaborations between industry and academia to foster the research. For instance, in February 2024, The National Cancer Institute (NCI) has announced that Washington University School of Medicine has joined a new clinical trials network to explore novel technologies that potentially identify different malignancies with a single blood test.
The clinical trial investigative site network market in Canada is witnessing significant growth due to smooth regulatory approvals and speedy process. For instance, in August 2024, Health Canada granted Ocugen, Inc. with a no objection letter to begin the OCU400 Phase 3 liMeliGhT clinical trial in Canada. It is one of the potential modifying gene therapy treatments for retinitis pigmentosa (RP) is OCU400. The clinical trial's expansion to Canada is significant because it provided researchers the chance to reach a larger patient base that includes a variety of gene variants linked to RP.
Asia Pacific Market Analysis
The clinical trial investigative site network market in Asia Pacific is exponentially expanding which can be ascribed to the clinical research field's ongoing development and expansion. In addition, trial management and operational efficiency are being enhanced by the expanding number of outsourcing companies. Technological developments like data analytics and digital health technologies are revolutionizing trial conduct by improving patient engagement and facilitating real-time monitoring.
The market in India is expecting substantial growth due to the governmental robust push towards innovating drug development. For instance, in August 2024, the first dengue vaccine phase 3 clinical trial in India was started by ICMR and Panacea Biotec using the indigenous Dengue vaccine, DengiAll. This phase 3clinical trial displayed country’s dedication to safeguard its people from the widespread illness and highlights India's prowess in vaccine development.
The clinical trial investigative site network market in China is witnessing lucrative growth opportunities dur to the fostering strategic alliances and partnerships. For instance, in October 2023, GSK plc announced that it and Chongqing Zhifei Biological Products, Ltd. (Zhifei) have reached an exclusive agreement to co-promote GSK's shingles vaccine, Shingrix, in China for a three-year initial period. The strategic alliance greatly increased Shingrix's availability by combining the size and experience of the two businesses, facilitating the quick growth of patient access to the vaccine and its potential future uses in the country.
Key Clinical Trial Investigative Site Network Market Players:
- Access Clinical Research
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- FOMAT Medical Research, Inc.
- SGS
- ICON Plc
- Meridian Clinical Research
- IQVIA Inc.
- Clinedge
- WCG
- ClinChoice
- KV Clinical
- SMO-Pharmina
- Xylem Clinical Research
- Aurum Clinical Research
The clinical trial investigative site network market is driven by the continuous innovations and breakthroughs by companies competing within this landscape. For instance, in June 2024, IQVIA Inc. introduced One Home, a new clinical trial technology platform. With a single sign-on and dashboard for several tasks and platforms, this platform is intended to simplify trial management. This, thus gives companies to bolster their capacities and expertise in providing efficacy in drugs and treatments portfolios.
Here's the list of some key players:
Recent Developments
- In August 2024, WCG acquired Array through the provision of extensive training materials that aid in the creation of qualified experts in the field of clinical research. This acquisition intended to improve WCG's services and portfolio.
- In March 2024, Cybin Inc., announced a successful end-of-phase 2 meeting for CYB003, its deuterated psilocybin analog, intended to treat major depressive disorder (MDD) as an adjuvant.
- Report ID: 7431
- Published Date: Aug 12, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Clinical Trial Investigative Site Network Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




