Antibody Therapeutics Market Outlook:
Antibody Therapeutics Market size was over USD 303.82 billion in 2025 and is projected to reach USD 1.04 trillion by 2035, witnessing around 13.1% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of antibody therapeutics is evaluated at USD 339.64 billion.

The expansion of the antibody therapeutics market is majorly driven by the rising demand for advanced therapies fueled by the increasing prevalence of chronic diseases such as cancer, autoimmune disorders, and infectious diseases. According to WHO report in December 2024, 43 million people led to death due to chronic diseases in 2021, which underscores the necessity of antibody therapeutics, further driving market expansion. Therefore, antibody-based treatments such as monoclonal antibodies, antibody-drug conjugates, and be-specific antibodies offer high specificity with low side effects, making them ideal to choose.
Additionally, the approvals of bispecific antibodies (BsAbs), which are a specialized type of therapeutic antibody, represent a breakthrough in immunotherapy, offering enhanced efficacy by targeting two different antigens simultaneously, thus driving the market. For instance, a study by the FDA in February 2024 highlighted the approvals from 2014 – 2023, which play a crucial role in expanding treatment options for conditions such as leukemia, lymphoma, and solid tumors. With such ongoing innovations and approvals, the market is anticipated to expand further in the forecast timeline.
FDA-approved be-specific antibodies
|
Trade Name |
Year Approved |
Indication |
|
Blincyto |
2014 |
Philadelphia chromosome-negative relapsed or refractory B cell precursor acute lymphoblastic leukemia. |
|
Hemlibra |
2017 |
Prevention of or reduction of bleeding episodes in hemophilia A with factor VIII inhibitors. |
|
Rybrevant |
2021 |
Locally advanced or metastatic non-small cell lung cancer with certain mutations. |
|
Kimmtrak |
2022 |
Unresectable or metastatic uveal melanoma. |
|
Vabysmo |
2022 |
Neovascular age-related macular degeneration and diabetic macular edema. |
|
Tecvayli |
2022 |
Relapsed or refractory multiple myeloma. |
|
Lunsumio |
2022 |
Relapsed or refractory follicular lymphoma. |
|
Epkinly |
2023 |
Relapsed or refractory diffuse large B-cell lymphoma. |
|
Columbia |
2023 |
Relapsed or refractory diffuse large B-cell lymphoma or large B-cell lymphoma. |
Source: FDA February 2024
Key Antibody Therapeutics Market Insights Summary:
Regional Highlights:
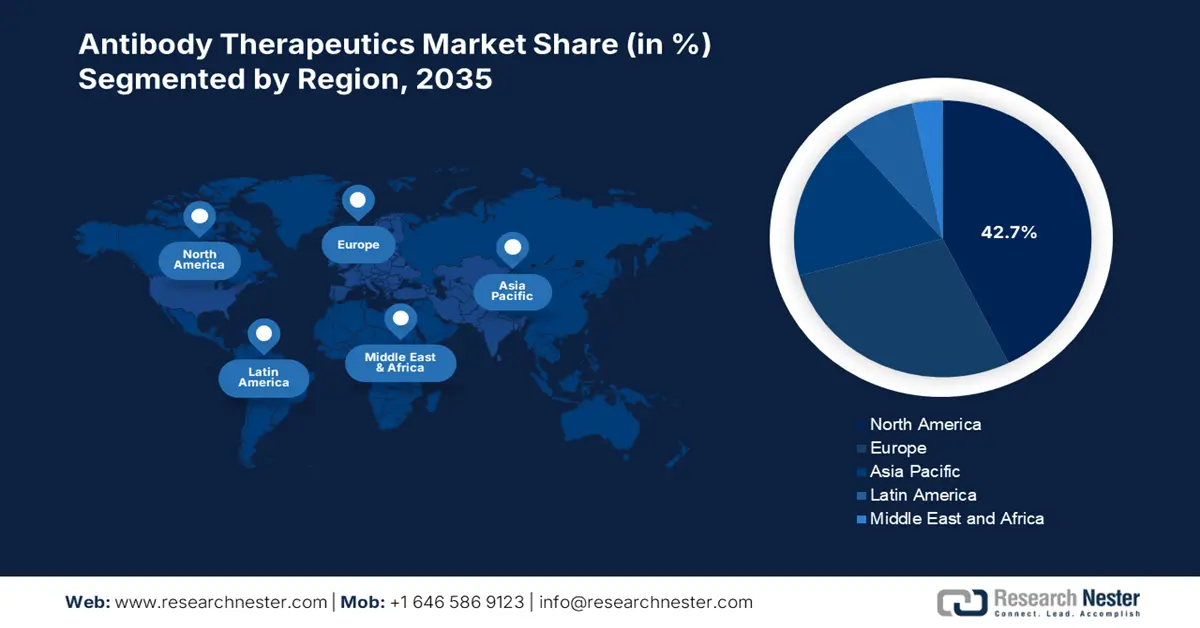
- North America holds a 42.7% share in the antibody therapeutics market, propelled by increased burden of diseases, pharmaceutical advancements, and government support, supporting strong growth prospects through 2035.
- The antibody therapeutics market in Asia Pacific is forecasted to achieve a significant CAGR by 2035, attributed to rapid growth of antibody clinical studies with strong investments.
Segment Insights:
- The Monoclonal Antibody segment is poised for substantial growth from 2026-2035, driven by its high efficacy and expanding applications in infections, carcinoma, and autoimmune disorders.
- The intravenous segment is projected to grow at a considerable rate from 2026 to 2035, driven by its fast action and effectiveness in delivering complex medications directly to the bloodstream.
Key Growth Trends:
- Increasing prevalence of chronic disorders
- Ongoing improvements in antibody engineering
Major Challenges:
- High cost and development issues
- Complex manufacturing and supply chain issues
- Key Players: AbbVie Inc., Johnson & Johnson, AbbVie Inc., Merck KGaA, Bristol-Myers Squibb, Regeneron Pharmaceuticals Inc..
Global Antibody Therapeutics Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 303.82 billion
- 2026 Market Size: USD 339.64 billion
- Projected Market Size: USD 1.04 trillion by 2035
- Growth Forecasts: 13.1% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (42.7% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, China, United Kingdom, Japan
- Emerging Countries: China, India, Japan, South Korea, Brazil
Last updated on : 12 August, 2025
Antibody Therapeutics Market Growth Drivers and Challenges:
Growth Drivers
- Increasing prevalence of chronic disorders: The rising occurrence of chronic diseases is driving the antibody therapeutics market as it boosts the demand for targeted therapeutics for the treatment procedure. Diseases such as autoimmune disorders, cancer, and inflammatory diseases require antibody therapeutics as they lack effective treatments. According to a report by the Autoimmune Institute in February 2024, autoimmune diseases affect 5% to 10% of the industrial world population, highlighting their low prevalence in developing countries. Therefore, this drives the antibody therapeutics market demand to treat such conditions with better patient outcomes.
- Ongoing improvements in antibody engineering: These advancements allow the antibodies to leverage their therapeutic properties. Through these technologies and methods, biotechnology and pharmaceutical firms can design antibodies with specific properties and applications. For instance, in December 2024, Parse Biosciences launched Evercode Mouse BCR to support antibody research in mice, which enables researchers to analyze the complete mouse BCR repertoire. Additionally, it improves antibody discovery, enhances workflows, reduces the need for additional sampling, and lowers research costs. Therefore, such advancements support innovation in the industry and upsurge the antibody therapeutics market globally.
Challenges
- High cost and development issues: Antibody therapeutics require extensive research, pre-clinical studies, and multi-phase clinical trials, often spanning over 10 to 15 years. Additionally, since these treatments are often personalized, regulatory approvals from authorities require comprehensive safety and efficacy while maintaining affordability, which is a complex challenge. Furthermore, small biotech firms often struggle to obtain funding while large pharmaceutical companies experience substantial financial risks due to higher chances of trial failures, depriving the market expansion.
- Complex manufacturing and supply chain issues: These complications occur due to the requirement of specialized bioprocessing facilities with stringent quality control measures. Unlike traditional drugs, antibody therapeutics are developed using living cells, making their manufacturing highly challenging and resource-intensive. Ensuring consistent quality and scaling up the production is difficult, as any minor variation can affect the efficacy of the final product. This makes the therapeutic production complex, thereby limiting accessibility and affordability for patients worldwide.
Antibody Therapeutics Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
CAGR |
13.1% |
|
Base Year Market Size (2025) |
USD 303.82 billion |
|
Forecast Year Market Size (2035) |
USD 1.04 trillion |
|
Regional Scope |
|
Antibody Therapeutics Market Segmentation:
Format (Monoclonal Antibody, Polyclonal Antibody Therapy, Bispecific Antibody, Antibody Fragment)
Based on format, the monoclonal antibody segment is set to hold antibody therapeutics market share of over 76.8% by the end of 2035. The domination is significantly influenced by its high efficacy and growing application for conditions such as infections, carcinoma, and autoimmune disorders. Additionally, the market expansion is further driven by industry developments enhancing monoclonal antibody production. For instance, in January 2025, Harbour BioMed with Sichuan Kelun Biotech BioPharmaceutical announced a license agreement with Windward Bio for HBM9378/SKB378, an anti-TSLP fully human antibody for immunological diseases. The deal includes exclusive rights for research, development, and commercialization, reflecting the growing market potential for monoclonal antibodies.
Route Of Administration (Intravenous, Subcutaneous)
Based on the route of administration, intravenous is projected to expand at a considerable rate during the forecast period in the antibody therapeutics market. The domination is due to its fast action, effectiveness, and ability to deliver complex medications directly to the bloodstream. The majority of the treatments require intravenous administration for an accurate dosage and quick results. In March 2022, Bristol Myers Squibb announced that the U.S. FDA approved the OpdualagTM fixed-dose dual immunotherapy combination for the treatment of unresectable or metastatic melanoma administrated as a single intravenous infusion. Therefore, it highlights the dominance of intravenous administration, ensuring precise delivery and enhanced efficacy.
Our in-depth analysis of the global market includes the following segments:
|
Format |
|
|
Disease Indication |
|
|
Route Of Administration
|
|
|
By Source |
|
|
End User |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Antibody Therapeutics Market Regional Analysis:
North America Market Analysis
North America in antibody therapeutics market is likely to account for more than 42.7% revenue share by the end of 2035, due to the increased burden of diseases, pharmaceutical advancements, and crucially, the government’s support, the industry is experiencing steady growth in the region. For instance, in October 2023, Celltrion USA, Inc. announced that the U.S. FDA approved ZYMFENTRA™ (infliximab-dyyb), which is the first and only subcutaneous infliximab for treating inflammatory bowel diseases. Thus, the approval strengthens market growth by enhancing treatment outcomes and driving innovation in monoclonal antibody drugs.
The antibody therapeutics market in the U.S. is witnessing significant growth due to the demand for antibody-based therapeutics and substantial investments by biotech organizations. For instance, in May 2021, AlivaMab Discovery Services, LLC announced the expansion of its drug discovery platform with the launch of a brand-new antibody engineering department led by Jonah Rainey, bringing 15 years of experience in antibody engineering. Such establishments enhance market growth through advancements in bispecific antibodies, strengthening clinical trials, and improving drug development, benefiting pharmaceutical and biotechnology companies.
Canada's market is growing steadily, supported by the government, a thriving biotech research ecosystem, and public-private collaborations. The country is focusing on enhancing the regulatory frameworks for faster approval, ensuring a competitive antibody therapeutics market landscape. In June 2021, AbCellera Biologics Inc. announced the construction of a state-of-the-art Good Manufacturing Practices (GMP) facility in Vancouver, Canada. The 130,000-square-foot facility, which is the first of its kind in the country, focuses on expanding AbCellera’s capabilities in developing and manufacturing antibodies. Such facilities will scale the market expansion by accelerating drug development and improving global pandemic preparedness.
APAC Market Statistics
Asia Pacific region houses a large and assorted population, including a substantial patient number from diseases that can be treated potentially through antibody therapies. This grants a considerable antibody therapeutics market prospect for increasing research and development investments and government initiatives supporting therapeutics, further driving market growth in the region. According to a report in March 2023 by Novotech Health Holdings Pte. Ltd, accounted for approximately 40% of global multi-specific antibody clinical trials between 2018 and 2022. The rapid growth of antibody clinical studies with strong investments will make it the fastest-growing region for antibody therapeutics.
India’s antibody therapeutics market is particularly supported by a robust biotech industry, growing biotech startups, and favorable regulatory reforms. With a focus on cost-effective biologic manufacturing, the country is anticipated to become a regional hub for antibody therapeutics. For instance, in February 2025, Equillium Inc. and Biocon Limited announced positive phase 2 study results for itolizumab in treating moderate to severe ulcerative colitis, leveraging a clinical remission rate of 23.3%. Such promising results strengthen the country’s position in the global market.
China's antibody therapeutics market is driven by a large patient base with unmet medical needs. The government’s commitment to supporting biotech funding, combined with favorable regulatory adjustments for faster approvals, has made pharmaceutical companies invest in therapeutics. In July 2023, Nona Biosciences announced a collaboration with Duality Biologics and BeiGene, Ltd., granting BeiGene an exclusive global clinical and commercial license for targeting solid tumors. Hence, the country’s antibody therapeutics market is anticipated to witness considerable growth during the forecast period by driving innovation.
Key Antibody Therapeutics Market Players:
- F. Hoffmann-La Roche Ltd.
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Developments
- Regional Presence
- SWOT Analysis
- AbbVie Inc.
- Johnson & Johnson
- AbbVie Inc.
- Merck KGaA
- Bristol-Myers Squibb
- Regeneron Pharmaceuticals Inc.
- Celsius Therapeutics, Inc.
- Novartis AG
- Amgen Inc.
- Biogen Inc.
- Ambrx Biopharma, Inc.
One of the key strategies adopted by companies in the antibody therapeutics market is investing in research and development and collaborations to enhance drug efficacy and expand their product portfolio. For instance, in March 2025, Harbour Biomed and AstraZeneca entered into a strategic collaboration to develop next-generation multispecific antibodies for immunology and oncology. The agreement includes a USD 105 million equity investment by AstraZeneca. Thus, the collaboration marks a significant investment in antibody therapeutics, further driving antibody therapeutics market growth.
Some of the prominent players are:
Recent Developments
In the News
- In June 2024, AbbVie Inc. announced the acquisition of Celsius Therapeutics, Inc. for USD 250 million in developing therapies for inflammatory diseases. This deal includes CE1383, an investigational antibody targeting TREM1 aimed at treating inflammatory bowel disease.
- In March 2024, Johnson & Johnson announced the acquisition of USD 2 billion of Ambrx Biopharma, Inc., enhancing its ADC development capabilities for targeted cancer therapies. The acquisition aims to advance next-generation ADCs, minimizing chemotherapy-related side effects.
- Report ID: 7414
- Published Date: Aug 12, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Antibody Therapeutics Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




