Anti-Inflammatory Biologics Market Outlook:
Anti-Inflammatory Biologics Market size was over USD 106.51 billion in 2025 and is anticipated to cross USD 196.21 billion by 2035, growing at more than 6.3% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of anti-inflammatory biologics is assessed at USD 112.55 billion.

Cases of inflammation in patients with conditions such as stroke, chronic respiratory disorder, heart disease, cancer, obesity, and diabetes have become more frequent than ever. According to a 2023 NLM report, 3 in every 5 habitats around the globe suffered from such conditions, and this population is enlarging with the heightening incidences of related illnesses. In this regard, a journal from The Lancet Rheumatology, published in October 2023, revealed that globally, over 17.6 million people were affected by rheumatoid arthritis (RA), showcasing a projection of 31·7 million by 2050. This is driving demand in the market to make required medicines more accessible for such residents seeking relief.
The utility of medicines from the anti-inflammatory biologics market majorly lies in their cost-effectiveness. This has converted these curatives into a first-line treatment approach for many healthcare settings, particularly in economically lagging regions. To display their payers’ pricing, many studies and systematic surveys have taken place. For instance, in the Journal of Biologic Meds, an article from October 2024 established a standard range of costs, USD 3,000.0-5,000.0, for these prescriptions related to asthma, allergies, eczema, and urticaria. It also marked the same for arthritis to be USD 10,000.0-30,000.0 per annum. It displays a variability in pricing, depending on the type of condition and other factors. Thus, companies are now focusing on producing more affordable options with wide applicability.
Key Anti-Inflammatory Biologics Market Insights Summary:
Regional Highlights:
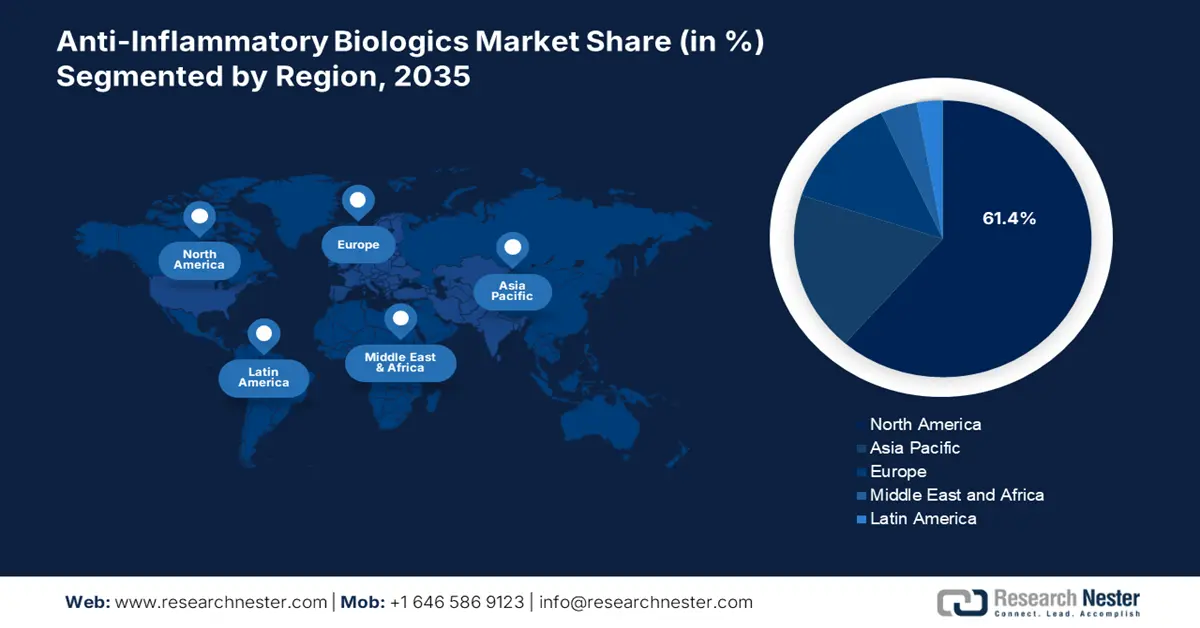
- North America commands a 61.00% share in the Anti-Inflammatory Biologics Market, with rising cases of arthritis and other inflammations increasing demand for biologic therapies, ensuring strong growth by 2035.
- Europe’s anti-inflammatory biologics market is expected to experience rapid growth through 2026–2035, driven by strong presence of global biopharmaceutical leaders and established biosimilar networks.
Segment Insights:
- The Rheumatoid Arthritis segment is expected to dominate market growth through 2026-2035, driven by an increasing aging population and higher prevalence of RA, as supported by WHO projections.
- The anti-tumor necrosis factor (TNF) segment is expected to hold a 45.7% share by 2035, driven by the rising prevalence of conditions like rheumatoid arthritis, psoriatic arthritis, and psoriasis.
Key Growth Trends:
- Advancements in therapeutic efficacy
- Expanding merchandise and application of biologics
Major Challenges:
- Limitations in large-scale production and distribution
- Growing burden of antimicrobial resistance
- Key Players: Novartis AG, AstraZeneca PLC., Pfizer Inc., Johnson & Johnson, UCB, Biocon Biologics Ltd., Sanofi.
Global Anti-Inflammatory Biologics Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 106.51 billion
- 2026 Market Size: USD 112.55 billion
- Projected Market Size: USD 196.21 billion by 2035
- Growth Forecasts: 6.3% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (61.4% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Japan, United Kingdom, France
- Emerging Countries: China, India, Brazil, Russia, Mexico
Last updated on : 12 August, 2025
Anti-Inflammatory Biologics Market Growth Drivers and Challenges:
Growth Drivers
- Advancements in therapeutic efficacy: In search of the most effective way of delivering therapies from the anti-inflammatory biologics market, many organizations have introduced innovative solutions. For instance, in February 2025, a team of researchers at the Institute of Nano Science and Technology (INST) Mohali created a self-actuating drug delivery system, nano micelles. It is a smart technology that allows the controlled release of medicinal agents, including biologics, right at the inflated joints in RA, offering better precision and safety. These discoveries signify the possible revolution in the clinical efficiency of these therapeutics, leveraging their acceptance worldwide and securing a prosperous future for this sector.
- Expanding merchandise and application of biologics: As per an estimation from the Biologic Meds Journal, the expenditure on these drugs accounted for 37.0% of the total drug spending in 2024. For instance, the 2023 list of regulatory-approved biologics for asthma from the Asthma and Allergy Foundation of America included NUCALA, CINQAIR, FASENRA, DUPIXENT, XOLAIR, and TEZSPIRE.
Challenges
- Limitations in large-scale production and distribution: Increasing accessibility in the anti-inflammatory biologics market requires bulk manufacturing and uninterrupted bioavailability. However, the complex process of formulation and the disrupted supply of bio-based reagents may bring variability and volatility to this sector. In addition, limited access to healthcare in underserved areas and stringent criteria of compliance often hinder its reach towards broader populations, restricting continuous expansion and worldwide adoption.
- Growing burden of antimicrobial resistance: The overuse of medicines from the anti-inflammatory biologics market is increasingly resulting in resistance. An NLM article from March 2024 exposed the high worldwide prevalence of such resistant cases for doripenem (97%), cefoxitin (96%), imipenem and cefuroxime (95%), ampicillin (94%), ceftriaxone (92%), aztreonam (91%), and meropenem (90%). This may hinder the traction of this sector, which needs extensive research, development, and funding to be mitigated.
Anti-Inflammatory Biologics Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
CAGR |
6.3% |
|
Base Year Market Size (2025) |
USD 106.51 billion |
|
Forecast Year Market Size (2035) |
USD 196.21 billion |
|
Regional Scope |
|
Anti-Inflammatory Biologics Market Segmentation:
Drug Class (Anti-tumor Necrosis Factor (TNF), Interleukins Antagonists, Janus Kinase (JAK) Inhibitor, Other)
In anti-inflammatory biologics market, anti-tumor necrosis factor (TNF) segment is poised to hold revenue share of over 45.7% by the end of 2035. This type of biologic has a wide spectrum of application including RA, psoriatic arthritis, ankylosing spondylitis, UC, CD, and psoriasis. Thus, the rising volume of the patient pools affected with these conditions, is solidifying the predominant captivity of this segment. For instance, in 2022, AbbVie secured a global net revenue of USD 21.2 billion for Humira (adalimumab), showcasing a 4.6% (worldwide) and 9.9% (in the U.S.) increment in the fourth quarter. Such strong market penetration, coupled with an internationally recognized safety profile, makes this segment the most revenue generating asset for this field.
Application (Rheumatoid Arthritis, Psoriasis, Others)
In terms of applications, the rheumatoid arthritis segment is projected to dominate the anti-inflammatory biologics market throughout the analyzed timeframe. This medical ailment refers to a higher prevalence and disability-adjusted life year (DALY) over other types of inflammatory disorders. According to NLM, RA contributed to 3,060,000 DALYs in 2020, with a notable prevalent onset at 60-70 years of age. Thus, the consumer base is magnifying with the worldwide aging population, where WHO projections confirmed the presence of 1.4 billion and 2.1 billion habitats aged 60 and over by 2030 and 2050, respectively. These figures testify to the continuous proprietorship of this sub-segment.
Our in-depth analysis of the global anti-inflammatory biologics market includes the following segments:
|
Drug Class |
|
|
Application |
|
|
Route of Administration |
|
|
Distribution Channel |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Anti-Inflammatory Biologics Market Regional Analysis:
North America Market Analysis
North America in anti-inflammatory biologics market is predicted to capture over 61.4% revenue share by 2035. There is significant demand for biologic therapies in this region due to the rising cases of arthritis and other inflammations. In a 2023 epidemiology, NLM highlighted that over 2.1 million American citizens were suffering from RA, while 50.0 million were being affected with chronic allergies every year. This is evidence of the increasing need for effective management therapeutics, inspiring global pioneers to supply for this surge. For instance, in July 2023, Biocon Biologics made its debut in this marketplace with the commercialization of Humira's biosimilar, HULIO (adalimumab-fkjp) injection.
The U.S. is augmenting the anti-inflammatory biologics market with its favorable regulatory framework. To combat the nationwide rampant of chronic inflammatory diseases, the governing authorities are actively cultivating medical resources through accelerated approvals. This is creating new business opportunities for both domestic and international giants. For instance, in September 2024, the FDA provided clearance to Johnson & Johnson for marketing its TREMFYA (guselkumab) in the U.S. market. This monoclonal antibody treats immune-mediated ulcerative colitis by targeting inflamed cytokines.
The biomanufacturing ecosystem of Canada, fueled by funds and subsidiary policies from the federal government, is promoting large-scale production in the anti-inflammatory biologics market. Learning from the pandemic strike has pushed the country to cultivate local resources of biologics to fight medical emergencies from inflammatory diseases. On this note, in a February 2025 article, the government of Canada highlighted the allocation of a total of USD 2.3 billion to 41 projects related to vaccines and therapeutics. The pan-Canadian network is distributed along Ontario, Quebec, Atlantic, Saskatchewan, Alberta, and British Columbia.
Europe Market Statistics
Europe is poised to witness the fastest growth in the anti-inflammatory biologics market by the end of 2035. The region holds predominant governance over biosimilars and generics, displaying an established network of accessible and affordable supply. This landscape is also supported by the strong presence of global biopharmaceutical leaders such as Sanofi, Novartis, Verona Pharma, GSK, and UCB. This is testified by the incremental sales of these therapeutics. For instance, in 2023, Sanofi revealed that the total full-year sales of Kevzara (sarilumab) accounted for USD 386.2 million, where USD 124.4 million alone was reported in Europe.
The UK is propagating in the anti-inflammatory biologics market with its capabilities in biopharmaceutical R&D and academic excellence. Research institutions across the country have been delivering the world with long-acting and safe alternatives for associated clinical illnesses. For instance, in November 2020, a team of scientists at the University of Oxford and the University of Birmingham conducted a multicenter, dose-ranging trial on otilimab for RA. A positive conclusion was established upon the study’s evaluation, presenting a notable reduction in inflammation. Such strong emphasis on clinical trials helps companies attain regulatory compliance.
Germany places a strong emphasis on manufacturing, making it a large production house for the anti-inflammatory biologics market. According to a report sponsored by the Federal Republic of Germany, the country generated USD 20.7 billion from the biopharmaceutical manufacturing industry in 2023, making it the 2nd largest producer in Europe in this discipline. In this cohort, the immunology segment, a crucial component in combating inflammation and autoimmune responses, exhibited a 9.0% growth rate from 2018 to 2023. Further, through commercial collaborations and promotional campaigns, the nation is strengthening its global presence in this sector.
Key Anti-Inflammatory Biologics Market Players:
- Amgen Inc.
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Novartis AG
- AstraZeneca PLC.
- Johnson & Johnson
- Pfizer Inc.
- GlaxoSmithKline plc
- F.Hoffmann-La Roche AG
- Merck & Co., Inc.
- Eli Lily and Company
- UCB
- Biocon Biologics Ltd.
- Regeneron Pharmaceuticals, Inc.
- Boehringer Ingelheim
- Sanofi
- Coherus BioSciences, Inc.
Key players in the anti-inflammatory biologics market are empowering their competency through global trades, R&D capacities, and efficiency in attaining maximum regulatory compliance. In this regard, the dominance of biosimilar producers has been observed throughout the emerging marketplaces. For instance, in August 2024, Biocon Biologics solidified the gateway of commercialization for its Bmab 1200 in Europe, the UK, Canada, and Japan. This was made possible by forming settlement and license partnerships with Janssen Biotech, Janssen Sciences Ireland, and Johnson & Johnson. Simultaneously, in March 2025, Regeneron Pharmaceuticals consolidated its footprint in Japan with the approval for its Dupixent (dupilumab) in treating COPD after its success in the U.S., China, and Europe. Such key players are:
Recent Developments
- In February 2025, Biocon Biologics made its debut in the U.S. biosimilar market by launching YESINTEK (ustekinumab-kfce). The biologic is available in 45 mg/0.5 mL PFS, 90 mg/mL PFS, 45 mg/0.5 mL vial, and 130 mg/26 mL vials, which are effective for several inflammatory conditions in an affordable way.
- In September 2024, UCB attained approval from the FDA for commercializing its BIMZELX (bimekizumab-bkzx). The monoclonal antibody is designed to treat active psoriatic arthritis, active non-radiographic axial spondyloarthritis with objective signs of inflammation, and active ankylosing spondylitis.
- Report ID: 7441
- Published Date: Aug 12, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Anti-Inflammatory Biologics Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




