Allergy Diagnostics and Therapeutics Market Outlook:
Allergy Diagnostics and Therapeutics Market size was over USD 38.12 billion in 2025 and is anticipated to cross USD 83.06 billion by 2035, witnessing more than 8.1% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of allergy diagnostics and therapeutics is assessed at USD 40.9 billion.

Allergy-based disorders are quite common worldwide and require a suitable diagnosis and an optimal treatment solution. According to the June 2024 Frontiers Organization report, a meta-analysis study revealed that the incidence rate of food allergy ranges from 3% to 35%. Therefore, the allergy diagnostics and therapeutics market gradually rotates around providing numerous testing services along with drug-based treatments for a varied array of allergies. The development of allergy usually takes place when a foreign substance develops in the immune system, resulting in the production of antibodies to liberate chemical elements such as histamine. Besides, the prevalence of allergies varies, depending on children and adults.
Allergy Morbidity Among Children and Adults
|
Allergy Type |
Children |
Adults |
|
Any allergy |
27.2% |
31.8% |
|
Seasonal allergy |
18.9% |
25.7% |
|
Eczema |
10.8% |
7.3% |
|
Food allergy |
5.8% |
6.2% |
Source: CDC February 2023
Furthermore, the allergy diagnostics and therapeutics market growth depends upon the management, evaluation, and accuracy of disease conditions. This further results in decision-making to select the appropriate test while considering the aspect of rising medical spending. In this regard, a clinical study published by NLM in July 2023 made a comparison of the payer’s pricing pertaining to the use of skin testing and sIgE testing as a part of allergy diagnosis. The study included a sample of 270 831 beneficiaries and 327 263allergy-related claims, comprising 34% of men and 66% of women. The cost of sIgE test was USD 15,903,954, USD 13,252,982 for intradermal tests, and USD 42,223,930 for skin prick tests, thereby enhancing the market demand.
Key Allergy Diagnostics and Therapeutics Market Insights Summary:
Regional Highlights:
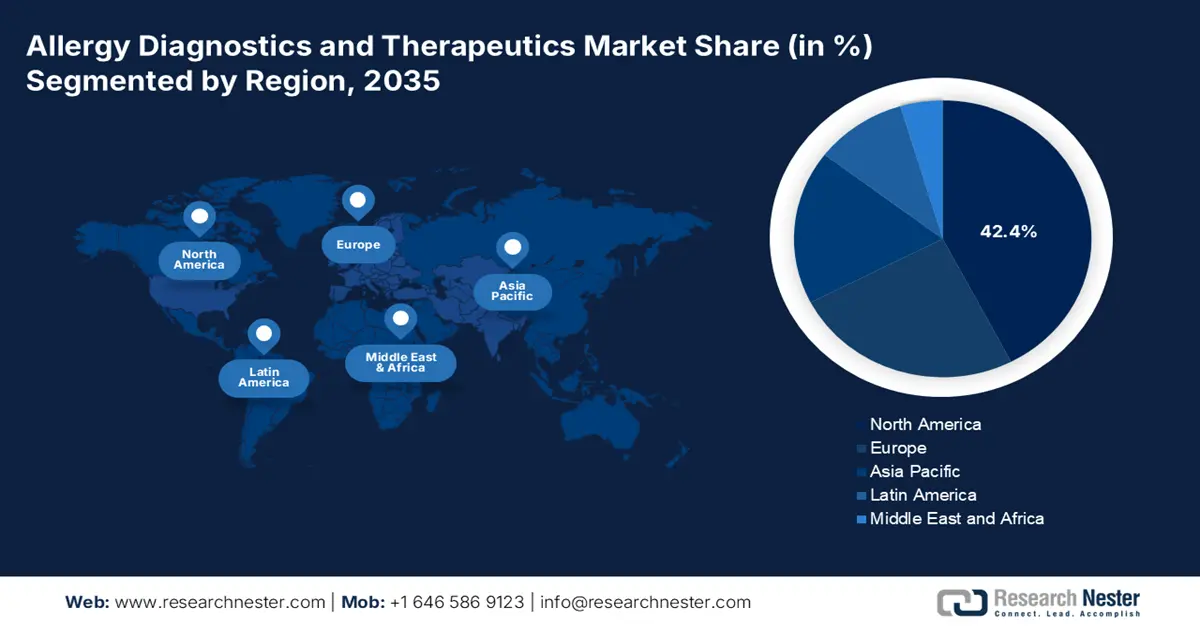
- North America’s 42.4% share in the Allergy Diagnostics and Therapeutics Market is driven by high incidence of respiratory allergic conditions and government strategies, reinforcing its market leadership through 2035.
- Asia Pacific's allergy diagnostics and therapeutics market is set for the fastest growth by 2035, fueled by urbanization, industrialization, and rising population.
Segment Insights:
- The Inhaled Allergen segment is projected to capture a 54.60% share by 2035, driven by increasing air pollution and industrial activities causing more inhaled allergies.
- The Consumables Segment is anticipated to hold a 67.8% share by 2035, propelled by rising allergy cases and demand for quick, convenient tests.
Key Growth Trends:
- Increased need for precision medicines
- Advancements and innovations in allergy therapies
Major Challenges:
- Deficit treatment solutions
- Unavailability of qualified health providers
- Key Players: DASIT Group SPA, bioMérieux, Thermo Fisher Scientific, Inc., Stallergenes Greer, Sun Pharmaceutical Industries Ltd.
Global Allergy Diagnostics and Therapeutics Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 38.12 billion
- 2026 Market Size: USD 40.9 billion
- Projected Market Size: USD 83.06 billion by 2035
- Growth Forecasts: 8.1% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (42.4% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Japan, United Kingdom, France
- Emerging Countries: China, India, Japan, South Korea, Singapore
Last updated on : 12 August, 2025
Allergy Diagnostics and Therapeutics Market Growth Drivers and Challenges:
Growth Drivers
-
Increased need for precision medicines: The shift towards the increasing adoption of personalized medicines is positively impacting the allergy diagnostics and therapeutics market globally. As per the February 2024 Personalized Medicine Coalition Organization report, precision medicines were positioned in 1/3rd of the latest drug approvals in 2023. In addition, the report notified the approval of 26 latest personalized treatment solutions, 19 prolonged suggestions for current personalized medicines, and 17 noteworthy extended indications for 12 diagnostic testing products. Therefore, with such developments, the market is expected to boost in the upcoming years.
-
Advancements and innovations in allergy therapies: The implementation of research and development activities has resulted in the development of cutting-edge therapy solutions which is another growth driver for the allergy diagnostics and therapeutics market. As per the February 2022 NLM article, allergen immunotherapy is one of the most novel therapies that comprises four forms- epicutaneous immunotherapy (EPIT), sublingual immunotherapy (SLIT), oral immunotherapy (OIT), and subcutaneous immunotherapy (SCIT). All these therapies are innovative in nature, thus amplifying market expansion across nations.
Challenges
-
Deficit treatment solutions: Despite the availability of effective therapies for a wide range of allergies, few allergies including food allergies comprise limited treatment options at present, hindering the allergy diagnostics and therapeutics market growth. The absence of decisive remedial treatments for precise allergies encounters patients. For instance, emergency epinephrine might be essential in unadorned cases of allergic reactions such as anaphylaxis. Epinephrine is frequently accessible as an auto-injector and can be lifesaving when managed promptly.
-
Unavailability of qualified health providers: The allergy diagnostics and therapeutics market is subjected to face restraints owing to the scarcity of educated and genuine medical professionals to cater to the condition. This eventually leads to probable issues in patient maintenance, admittance, and superiority, particularly in underdeveloped areas. According to the 2025 WHO report, there will be a shortfall of almost 11 million health workers by 2030, especially in low- and lower-middle-income nations. Therefore, with such a prediction, the market is expected to experience a drawback in terms of its growth.
Allergy Diagnostics and Therapeutics Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
CAGR |
8.1% |
|
Base Year Market Size (2025) |
USD 38.12 billion |
|
Forecast Year Market Size (2035) |
USD 83.06 billion |
|
Regional Scope |
|
Allergy Diagnostics and Therapeutics Market Segmentation:
Product (Consumables, Instruments, Services)
Consumables segment is expected to capture allergy diagnostics and therapeutics market share of over 67.8% by 2035. This is possible owing to the rising cases of allergies and the necessity for quick tests to classify allergic circumstances. Besides, the convenience of kits explicit to each allergen and the comfort of accessibility are other factors driving the growth of the segment. In July 2024, AliveDx announced the approval of IVDR CE mark for its revolutionary microarray immunoassay, designed for the recognition of a precise IgE directed to a protein allergen in human serum. Therefore, with such development and approval, the segment is expected to boost by the forecast period.
Allergen Type (Inhaled, Food, Drug)
In allergy diagnostics and therapeutics market, inhaled segment is set to capture revenue share of over 54.6% by 2035. Factors such as increasing air pollution across nations, cumulative construction activities, and chemical-based activities in industries are readily driving inhaled allergies. For instance, as per the January 2022 Allergology International article, 17.1% to 54.8% of the general population in Europe are positive for IgE to any inhaled allergen. This has eventually resulted in the occurrence of asthma, affecting both children and adults internationally with increased incidence rates.
Asthma Incidence Among Children and Adults
|
Characteristic |
Weighted Number |
Percentage |
|
Children |
4,675,475 |
6.5% |
|
Adults |
20,288,399 |
8.0% |
Source: CDC May 2023
Our in-depth analysis of the allergy diagnostics and therapeutics market includes the following segments:
|
Product |
|
|
Drug Class |
|
|
Test |
|
|
Allergen Type |
|
|
End User |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Allergy Diagnostics and Therapeutics Market Regional Analysis:
North America Market Analysis
North America’s allergy diagnostics and therapeutics market is expected to capture revenue share of over 42.4% by 2035. The high incidence of respiratory allergic havoc, promising government strategies, cumulative consciousness of allergy diagnostics, acquisition of in vitro blood tests, and technological progressions in the healthcare industry are a few factors driving the market in the region. According to the October 2024 CMS report, the regional healthcare expenditure expanded by 7.5% in 2023 and reached USD 4.9 trillion, accounting for USD 14,570 per person. Additionally, based on the gross domestic product, the spending constituted 17.6%, thus an optimistic outlook for the market in the region.
The allergy diagnostics and therapeutics market in the U.S. is witnessing significant growth owing to the presence of administrative organizations focusing on approvals of newly developed therapy solutions. For instance, in February 2024, the U.S. FDA approved Xolair (omalizumab) injection for immunoglobulin E-mediated food allergy among adults and children to reduce Type 1 allergic reactions. This further included the reduction of anaphylaxis that may happen with accidental acquaintance with one or more food intake. This injection is useful since 6% of the population in the country suffers from food allergies and exposure to certain food products that constitute life-threatening allergic reactions.
The allergy diagnostics and therapeutics market in Canada is gaining more exposure since the government has taken the initiative to make suitable investments to introduce programs to combat the condition. According to the 2025 Federal Budget, Food Allergy Canada and the Canada Society of Allergy and Clinical Immunology (CSACI) recommended a USD 4.5 million investment to implement the National Food Allergy Action Plan. The objective of this plan is to protect lives, diminish food allergy occurrence, reduce healthcare costs, lessen the economic influence, and expand the quality of life for the millions impacted within the country.
APAC Market Statistics
Asia Pacific is constituted to be the fastest-growing region, positively impacting the evolution of the allergy diagnostics and therapeutics market. The aspect of urbanization, industrialization, and rising population are readily driving market expansion in the region. According to the 2025 Asia and Pacific UNFPA Organization report, approximately 60% of the global population resides in this region with 4.3 billion people and populated countries such as India and China. In addition, organizations in the region are efficiently contributing towards market growth through service expansion, product innovation, and standard partnership deals to enhance the market demand.
In India, the presence of allergy treatment services ranges from symptom management to the provision of medications including corticosteroids and antihistamines, thus driving the allergy diagnostics and therapeutics market. In June 2024, Dr. Reddy’s Laboratories Ltd. signed a conclusive contract with Haleon plc to purchase shares of Northstar Switzerland SARL and obtain Haleon’s global range of consumer healthcare brands in the Nicotine Replacement Therapy (NRT). This acquisition collided with Dr. Reddy’s product portfolio for skincare, cough-cold-allergy, and hydration, thereby being fruitful for market expansion and demand.
China’s allergy diagnostics and therapeutics market is attributed by the creation of consensus that aims to regulate the diagnostic approaches of allergens and deliver an orientation for healthcare givers. Besides, as stated in the March 2021 NLM article, the occurrence of allergic-based illnesses in the country has affected the quality of life of approximately 40% of the total population. This has resulted in the formation of co-sensitization and cross-sensitization, both of which cause complications in allergy diagnostic services. Therefore, the implication of the consensus based on four aspects of allergy disorder including mechanism, diagnosis procedures, allergen detection, and distribution, the market is expected to uplift in the country.
Key Allergy Diagnostics and Therapeutics Market Players:
- AbbVie, Inc.
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- R-Biopharm AG
- EUROIMMUN Medizinische Labordiagnostika AG
- DASIT Group SPA
- bioMérieux
- Thermo Fisher Scientific, Inc.
- Stallergenes Greer
- Sun Pharmaceutical Industries Ltd
- Minaris Medical America, Inc.
- Siemens Healthcare GmbH
- AESKU.GROUP GmbH
- Omega Diagnostics Group PLC
- HYCOR Biomedical
- Lincoln Diagnostics, Inc.
- Alcon
- HOB Biotech Group Corp., Ltd.
- Danaher
- Allergy Therapeutics
- Sanofi
- Pfizer, Inc.
- Teva Pharmaceutical Industries Ltd.
- GSK plc
- F. Hoffmann-La Roche Ltd.
- Merck & Co., Inc.
- ARS Pharmaceuticals, Inc.
- Camallergy
- OnDosis
The allergy diagnostics and therapeutics market is extremely competitive and key companies are involved in activities such as tactical collaborations, latest product introductions, procurements, technological advancements, and regional growth. For instance, in November 2022, Allergy Partners declared a deliberate collaboration with Aimmune Therapeutics, Inc. The purpose of this collaboration was to provide Allergy Partners’ patients ranging between 4 years and 17 years with a long-established diagnosis of peanut allergy with straight access to PALFORZIA, the first and only FDA-approved peanut allergy treatment. Therefore, this denotes an optimistic outlook for the market to expand globally.
Here's the list of some key players:
Recent Developments
- In November 2024, ARS Pharmaceuticals, Inc. announced a licensing agreement with ALK-Abelló A/S to provide ALK restricted rights to commercialize neffy, the first and only needle-free emergency treatment for Type I allergic reactions including anaphylaxis, in Europe, Canada, and other locations outside of the U.S.
- In November 2023, Camallergy and OnDosis notified about their collaboration to alter the way food allergies are treated by combining Camallergy's groundbreaking therapies with OnDosis' cutting-edge Dosage Manager device technology.
- Report ID: 7418
- Published Date: Aug 12, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Allergy Diagnostics and Therapeutics Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




