Intragastric Balloon Market Outlook:
Intragastric Balloon Market size was over USD 60.5 million in 2025 and is poised to exceed USD 196.46 million by 2035, witnessing over 12.5% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of intragastric balloon is evaluated at USD 67.31 million.

Intragastric balloon market growth has positive trends that are driven by the synergy between increasing obesity worldwide. For instance, World Heart Federation unveiled that as per World Obesity Federation's 2022 World Obesity Atlas, one billion people worldwide, including one in five women and one in seven men will suffer from obesity by 2030. Furthermore, the availability of less invasive weight loss methods is becoming more widely known to the patient population and healthcare professionals due to its effectiveness. For instance, in October 2024, a study by the National Institutes of Health demonstrated that participants involved in the study experienced an average 20% decrease in body mass index (BMI), 55% in type 2 diabetes, 57% in hypertension, and 54% in abnormal cholesterol after ten years.
In addition, balloon technology and biocompatible material represent a dominant growth driver for improved therapeutic impact along with tolerability by patients. For instance, in March 2024, through an endoscope, MIT has created a new gel called GastroShield. It can be sprayed onto the surgical sites in order to protect the damaged area, this gel creates a strong yet pliable layer. Moreover, market trends foresee a trend to be anticipated away from intricate balloon systems, such as adjustable and ingestible modalities, that provide higher patient customization and procedural convenience. These synergistic combinations place the intragastric balloon as a strategically critical therapeutic modality in the emerging weight management paradigm.
Key Intragastric Balloon Market Insights Summary:
Regional Highlights:
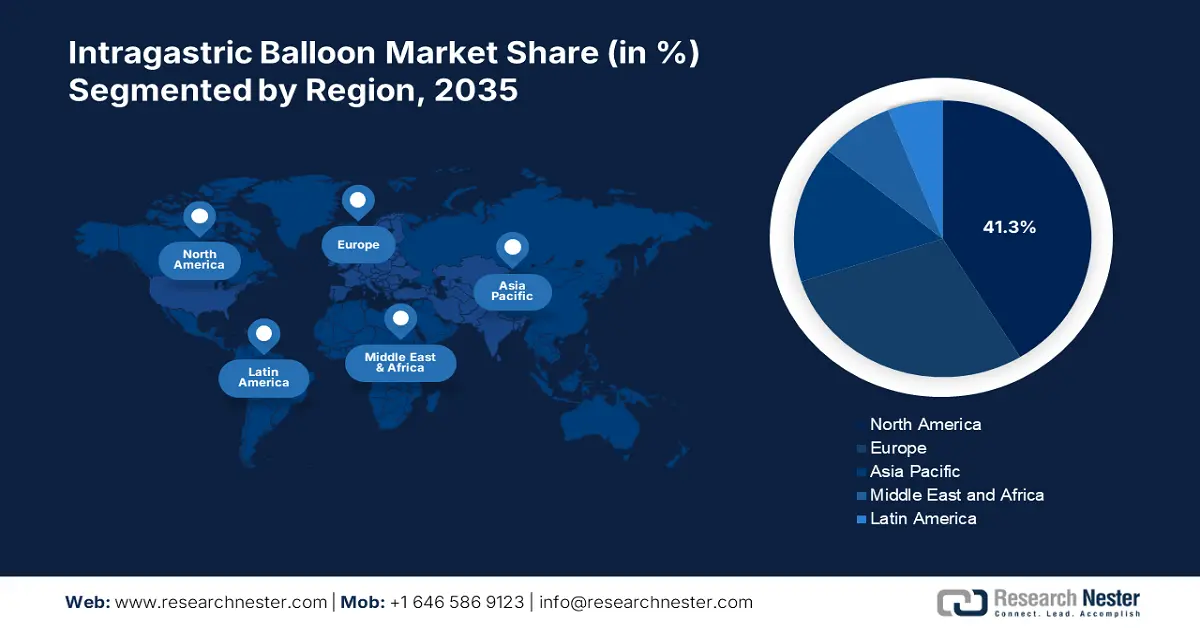
- North America intragastric balloon market will hold around 41.30% share by 2035, driven by a high rate of obesity, advanced medical infrastructure, and well-established reimbursement streams.
- Asia Pacific market projects lucrative growth during the forecast timeline, driven by the rising middle-class population, increasing lifestyle diseases, and greater access to advanced medical equipment.
Segment Insights:
- The endoscopy segment in the intragastric balloon market is expected to achieve a 75.40% share by 2035, attributed to its established safety profile and procedural comfort with gastroenterologists.
- The saline filled segment in the intragastric balloon market is poised to achieve a major share by 2035, fueled by its proven safety profile and reliable deflation behavior.
Key Growth Trends:
- Demand for minimally invasive procedures
- Integration with lifestyle modifications program
Major Challenges:
- Patient tolerability and side effects
- Procedural complications
Key Players: Allurion Technologies, Allergan (Acquired by Abbott), Apollo Endosurgery, Reshape Medical, Spatz FGIA, Endalis, Silimed, Medsil.
Global Intragastric Balloon Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 60.5 million
- 2026 Market Size: USD 67.31 million
- Projected Market Size: USD 196.46 million by 2035
- Growth Forecasts: 12.5% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (41.3% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Japan, China, France
- Emerging Countries: China, India, Japan, Brazil, Mexico
Last updated on : 8 September, 2025
Intragastric Balloon Market Growth Drivers and Challenges:
Growth Drivers
- Demand for minimally invasive procedures: Growing patient demand for less invasive treatment is expanding the market owing to the fact that patients have a need to minimize risk for surgery, recovery time, and pain after treatment. For instance, in June 2024, for patients with gastric cancer, Massachusetts General Hospital developed a new technique that offers minimally invasive gastrectomy by utilizing the robotic instruments' enhanced range of motion and surgical precision. Therefore, the intragastric balloon as an endoscopic, non-surgical treatment meets patient demand since it provides a relatively less invasive method of treatment compared to conventional bariatric treatments.
- Integration with lifestyle modifications program: The combination of adding lifestyle changes with effective health programs spurs the market growth. This two-barreled strategy provides maximum effect from the balloon and helps towards complete weight control and therefore increase patient outcomes and decrease recidivism. For instance, in March 2025, to combat the developing obesity rates and gastric issues, the India government has initiated programs such as the Fit India Movement, NP-NCD, POSHAN Abhiyaan, Eat Right India, and Khelo India. In order to ensure a healthier future for everybody, these programs seek to promote long-term behavioral change.
Challenges
- Patient tolerability and side effects: The most challenging hurdle for the market is the physiological response of the body. Side effects may be temporary, but they may have the potential to lead to premature balloon removal and reduced patient compliance and therefore reduce overall therapeutic efficacy and penetration into the market. Minimization, therefore, of such reactions by changing balloons, pharmacologic management of conditions, and promotion of patient care standards still remains a goal in restricting this condition and achieving maximum patient acceptability.
- Procedural complications: The growth of intragastric balloon market is hampered through endoscopic balloon placement and removal as they are not risk-free involving some risk in the form of perforation of the stomach, migration causing intestinal obstruction, and deflation of the stomach. These serious adverse events, though uncommon, necessitate strict patient selection criteria, uniform procedural method, and prompt availability of specialized care. Prevention of such complications, by technological progress and improved physician education, continues to be of the utmost priority in the interest of patient safety and upholding the integrity of intragastric balloon therapy.
Intragastric Balloon Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
CAGR |
12.5% |
|
Base Year Market Size (2025) |
USD 60.5 million |
|
Forecast Year Market Size (2035) |
USD 196.46 million |
|
Regional Scope |
|
Intragastric Balloon Market Segmentation:
Administration Segment Analysis
Endoscopy segment is anticipated to account for intragastric balloon market share of around 75.4% by 2035, owing to its established safety profile and procedure comfort with gastroenterologists. The procedure provides accurate placement and removal of the balloon under direct vision, reducing the rate of complications over other administration routes. For instance, in May 2024, the FDA approved the new EVIS X1 endoscopy system and two compatible gastrointestinal endoscopes, the CF-HQ1100DL/I colonovideoscope and the GIF-1100 gastrointestinal videoscope, which is approved for use in the upper digestive tract, which includes the esophagus, stomach, and duodenum.
Filling Material Segment Analysis
Based on the material, the saline-filled valves segment is expected to garner the major share in the intragastric balloon market by the end of 2035 attributable to its proven safety profile and reliable deflation behavior. For instance, in October 2023, following a nationwide clinical trial being supervised by the MD professor of gastroenterology at the University of Colorado School of Medicine and director of the Metabolic and Bariatric Program, the FDA approved a new weight-loss device. A swallowable gastric balloon, Allurion Balloon doesn't require invasive procedures. Saline is a biocompatible substance that reduces the risk of reaction and allows easy balloon volume adjustment. Such proven track record with long clinical history of saline-filled devices contributes to physicians' and patients' faith.
Our in-depth analysis of the global market includes the following segments:
|
Balloon Type |
|
|
Filling Material |
|
|
Administration |
|
|
End use |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Intragastric Balloon Market Regional Analysis:
North America Market Insights
North America intragastric balloon market is expected to capture revenue share of over 41.3% by 2035, driven by its high rate of obesity, thereby providing an enormous and easily accessible pool of patients. This common clinical requirement, combined with the region's highly advanced medical infrastructure and well-established reimbursement streams, enables rapid take-up and dissemination of intragastric balloon treatments. Thus, sheer numbers of prospective patients stimulates the surging demand for market.
The U.S. market is growing exponentially owing to the smooth approvals and conducive regulatory ecosystem that push innovation & breakthroughs in gastric balloon techniques. For instance, in April 2022, it was revealed that the FDA authorized the Spatz3, the first adjustable intragastric balloon (aIGB) on the market in the United States, in October 2021. For individuals with obesity who have not been able to lose weight and keep it off with a supervised weight control program, it is recommended for short-term use (up to eight months).
Canada market is likely to witness substantial growth during the forecast period driven by its universal health policy, offering comparatively extended access to weight reduction interventions, such as intragastric balloons, when medically necessary. For instance, in November 2024, Novo Nordisk's new weight-loss medication, Wegovy has been licensed by Canada's health regulator to lower the risk of myocardial infarction or nonfatal heart attack in adults. It was the first medication authorized in Canada to treat adults with existing cardiovascular disease who are obese and at risk for heart-related diseases.
Asia Pacific Market Insights
The intragastric balloon market in Asia Pacific is gaining traction and is expected to witness lucrative growth during the forecast timeline. The region offers a fast-growing environment in the market, mainly because of the middle-class growth and the subsequent increase in lifestyle diseases, with obesity and metabolic syndrome at the forefront. This demographic change, combined with greater access to sophisticated medical equipment and greater awareness of minimally invasive weight loss procedures, presents a huge and growing patient base.
India increasing market is being driven foremost by the rapidly expanding urban population of the nation and the parallel rise in sedentary living, thereby recording a fast boost in obesity as well as connected comorbidities. For instance, in March 2025, according to data from the National Family Health Survey (NFHS)-5 (2019–21), 24% of Indian women and 23% of Indian men are overweight or obese. 6.4% of women and 4.0% of men in the 15–49 age group are obese, according to the NFHS-5 (2019–2021). Additionally, the proportion of children under five who are overweight (weight-for-height) has increased from 2.1 percent in NFHS-4 (2015–16) to 3.4 percent in NFHS-5 (2019–21) at the All-India level.
China intragastric balloon market is witnessing lucrative growth opportunities owing to its robust technological-driven ecosystem, which fosters evolution in technologies and methodologies for treatments. For instance, in February 2025, the launch of the high-end precision minimally invasive surgical tools production expansion project was launched in Zibo City. Xinhua Surgical Instruments Co., Ltd., announced with pride an anticipation of more than 4.7 million pieces of open surgical instruments and 19,000 sets of minimally invasive laparoscopic surgical instruments to be produced annually. This represents a significant increase in production capacity and will firmly establish the company's position in the market.
Intragastric Balloon Market Players:
- Allurion Technologies
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Allergan (Acquired by Abbott)
- Apollo Endosurgery
- Reshape Medical
- Spatz FGIA
- Endalis
- Silimed
- Medsil
The company landscape in the intragastric balloon market is predominantly driven by the ecosystem that involve relentless clinical trials and innovations in overcoming the gastric diseases and problems. For instance, in November 2024, MariTide is an experimental antibody peptide conjugate subcutaneously injected monthly or less frequently, is the subject of a double-blind, dose-ranging Phase 2 research that Amgen reported positive results at 52 weeks. It showed up to 20% average weight loss at week 52 without a weight loss plateau in individuals with obesity or overweight who did not have Type 2 diabetes, suggesting the possibility of additional weight loss after 52 weeks.
Here's the list of some key players:
Recent Developments
- In December 2024, Boston Scientific introduced the Apollo ESG System has been approved by the US FDA and CDSCO. It is a safe, clinically successful substitute and a minimally invasive weight loss technique.
- In June 2023, Allurion partnered with Medtronic in order to expand its AI-powered weight loss program throughout Central and Eastern Europe as well as the Middle East and Africa (CEMA).
- Report ID: 1431
- Published Date: Sep 08, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Intragastric Balloon Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




