Intracranial Pressure Monitoring Devices Market Outlook:
Intracranial Pressure Monitoring Devices Market size was over USD 1.8 billion in 2025 and is projected to reach USD 3.64 billion by 2035, witnessing around 7.3% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of intracranial pressure monitoring devices is evaluated at USD 1.92 billion.

The implementation of intracranial pressure monitoring devices constitutes the use of consumables and accessories relevant to measuring and monitoring the pressure inside the skull. As per the January 2024 NLM article, a usual intracranial pressure (ICP) device ranges from 7mm to 15 mm Hg, while it does not surpass 15 mm Hg in the vertical position. Besides, ICP lowering therapy operates when pressure exceeds 20mm to 25 mm Hg, suitable for administering and evaluating brain functionality. Additionally, ICP monitoring permits the cautious use of intercessions with a distinct target point, thereby evading possibly harmful aggressive treatment and effectively driving the market globally.
Moreover, the functionality and accessibility of ICP monitoring devices hugely depend upon the presence of integrated circuits that ensure signal and telemetry processing. Technologies such as microchips and resonant circuits are highly utilized to translate pressure variations into quantifiable signals. In this regard, the 2023 OEC report has stated the trading-based statistics of integrated circuits that positively impact the market. The global trade valuation of these circuits is USD 910 billion with Taipei being the top exported at USD 214 billion and Hong Kong as the top importer at USD 200 billion. Also, the product complexity is 1.2 and it is the fourth most traded product internationally.
Nationwide Integrated Circuits Export/Import
|
Countries |
Export |
Import |
|
Taipei |
USD 214 billion |
USD 60.3 billion |
|
China |
USD 154 billion |
USD 200 billion |
|
South Korea |
USD 134 billion |
USD 49.7 billion |
|
Malaysia |
USD 79.3 billion |
- |
|
Singapore |
USD 64.2 billion |
- |
|
Hong Kong |
USD 200 billion |
- |
Source: OED 2023
Key Intracranial Pressure Monitoring Devices Market Insights Summary:
Regional Highlights:
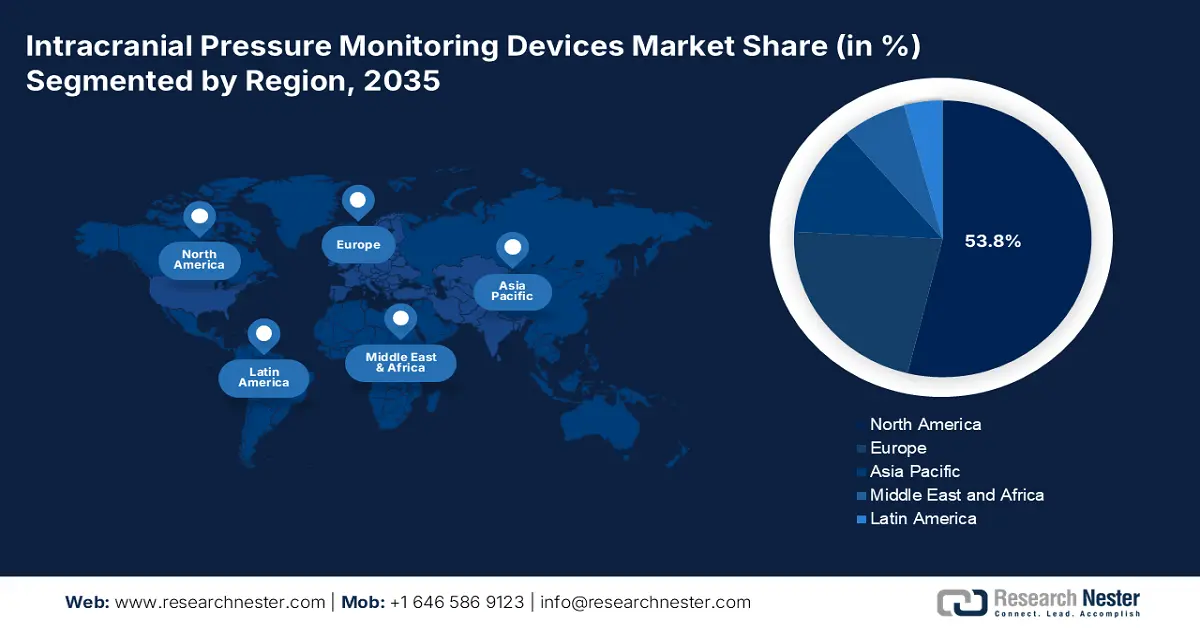
- North America dominates the Intracranial Pressure Monitoring Devices Market with a 53.80% share, driven by high incidence of brain injuries, strong reimbursement, and key players, fostering growth through 2035.
- Asia Pacific's intracranial pressure monitoring devices market is set for lucrative growth by 2035, fueled by rising trauma and sports injuries in the region.
Segment Insights:
- The Invasive Intracranial Pressure Monitoring Devices segment is expected to hold 94.2% market share by 2035, propelled by the need for precise monitoring through direct sensor or catheter placement.
- The Traumatic Brain Injury segment is anticipated to achieve lucrative growth through 2035, driven by the rising incidence of head trauma and demand for timely diagnosis.
Key Growth Trends:
- Increased adoption of wireless monitoring devices
- Prevalence of remote monitoring
Major Challenges:
- High pricing and inadequate repayment strategies
- Shortage of skilled professionals
- Key Players: Codman & Shurtleff Inc., RAUMEDIC AG, Vittamed, Sophysa, Orsan Medical Technologies.
Global Intracranial Pressure Monitoring Devices Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 1.8 billion
- 2026 Market Size: USD 1.92 billion
- Projected Market Size: USD 3.64 billion by 2035
- Growth Forecasts: 7.3% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (53.8% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Japan, United Kingdom, France
- Emerging Countries: China, India, Brazil, Russia, Mexico
Last updated on : 12 August, 2025
Intracranial Pressure Monitoring Devices Market Growth Drivers and Challenges:
Growth Drivers
- Increased adoption of wireless monitoring devices: These devices offer patients better flexibility and convenience while dipping the burden on healthcare facilities. Additionally, organizations capitalize on this trend by evolving devices that are easy to use and offer consistent and precise data. For instance, in November 2023, Worldsensing launched its latest wireless sensor, The Vibration Meter, which measures vibrations in long-term and nonstop vibration monitoring projects. Therefore, these devices are readily increasing which positively impacts the market globally.
- Prevalence of remote monitoring: The intracranial pressure (ICP) monitoring devices market growth is attributed to the increasing manufacturing of devices utilized for remote monitoring. In addition, this has increased the adoption of telehealthcare and telemedicine services across all nations. According to the December 2024 report, the increased use of telehealth services resulted in the medicare economic index of 3.5% in 2025 with a facility charge of USD 31.0. Based on this, there is the continuous development of advanced and innovative technologies to ensure instantaneous functioning and data processing, thus a positive outlook for the market.
Challenges
-
High pricing and inadequate repayment strategies: ICP monitoring devices are usually expensive, which creates a barrier to their acceptance in a few counties or by patient inhabitants. Moreover, restricted or insufficient compensation guidelines also limit the mandate for these devices, thus hindering the expansion of the market internationally. Besides, both these factors restrain revolution, and accessibility to care, possibly leading to monetary tension for both patients and healthcare providers.
- Shortage of skilled professionals: This is another factor negatively impacting the market globally. These monitoring devices involve the placement of a catheter inside the brain, which can be hazardous and may lead to difficulties, resulting in a minimal acceptance rate. The appropriate use and clarification of intracranial pressure monitoring devices necessitate dedicated and focused training, but with the presence of poorly skilled professionals, the market is subjected to face challenges that affect its growth.
Intracranial Pressure Monitoring Devices Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
CAGR |
7.3% |
|
Base Year Market Size (2025) |
USD 1.8 billion |
|
Forecast Year Market Size (2035) |
USD 3.64 billion |
|
Regional Scope |
|
Intracranial Pressure Monitoring Devices Market Segmentation:
Product (Invasive, Non-Invasive)
Invasive intracranial pressure monitoring devices market is expected to capture revenue share of over 94.2% by 2035. To evaluate intracranial pressure, invasive machinery frequently involves the installment of a catheter or sensor straight inside the cerebrospinal fluid. This ensures precise monitoring that includes intrusive procedures. In June 2023, Luciole Medical AG notified the acquisition of Spiegelberg GmbH & Co. KG from SHS Capital. Through this acquisition, Spiegelberg’s recognized ICP monitors complement Luciole’s minimally invasive brain monitoring solutions, thereby a suitable opportunity for the segment’s growth.
Application (Traumatic Brain Injury, Intracerebral Hemorrhage, Meningitis, Subarachnoid Hemorrhage, CSF Management, Migraine, Stroke, Hydrocephalus, EEG)
Based on the application, the traumatic brain injury segment is expected to garner a lucrative share in the intracranial pressure (ICP) monitoring devices market by the end of 2035. This category of injury is a common application for intracranial pressure monitoring devices since intracranial pressure is a mutual problem in patients. According to the 2025 Centre for Neuro Skills report, the average incidence rate of the condition is 95 per 100,000 population, out of which 22% expires from the injuries caused. This is quite common among adolescents, young adults, and people older than 75 years of age. Therefore, to combat this and provide suitable treatment, there is a huge demand for the market internationally.
Our in-depth analysis of the global market includes the following segments:
|
Product |
|
|
Application |
|
|
End User |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Intracranial Pressure Monitoring Devices Market Regional Analysis:
North America Market Analysis
North America intracranial pressure monitoring devices market is poised to dominate over 53.8% revenue share by 2035. The region’s supremacy is accredited to factors including a high prevalence of distressing brain damage, the existence of foremost market players, and satisfactory reimbursement policies. Besides, the rising prevalence of migraine in the region is also enhancing the demand of the market. As per the May 2024 NLM article, migraine has severely affected the population with an incidence rate ranging from 11.7% to 14.7%, thereby a positive outlook for market upliftment.
The intracranial pressure (ICP) monitoring devices market in the U.S. is rising exponentially owing to the presence of robust medical infrastructure, rising healthcare spending, government initiatives, and the adoption of neurostimulation devices. According to the December 2024 CMS report, the regional health spending was USD 4.9 trillion, denoting an increase of 7.5% as of 2023. Therefore, this allows investment strategies by the government to provide increasing funds for the development of technology and facilities in the country. Additionally, the establishment of the America Society of Craniofacial Surgery (ASCFS), crucial for creating awareness, is also a driving factor for the market in the country.
The intracranial pressure monitoring devices market in Canada is likely to witness substantial growth during the forecast period driven by the introduction of the latest innovative monitoring devices. For instance, in November 2024, Medical Device Network unveiled Aktiia’s introduction of its groundbreaking blood pressure monitoring wearables in the country. This includes the launch of a continuous blood pressure monitoring (CBPM) bracelet after securing Health Canada approval. This is an effective contribution to the market as well as catering to the health of the population through standard monitoring capabilities.
APAC Market Statistics
The intracranial pressure monitoring devices market in Asia Pacific is gaining a positive influence and is projected to witness lucrative growth during the forecast timeline. Factors such as increasing incidences of trauma and sports injury are highly responsible for the expansion of the market within the region. For instance, as per the February 2021 NLM article, a clinical study was conducted on 782 athletes in Korea, wherein 141 injuries were recorded and 15.4% of athletes were severely suffering from injuries. Besides, 71.4% of injuries occurred among sport-climbing athletes, thereby increasing the exposure of the market in the region.
The market in India is highly driven by the availability of both invasive and non-invasive device technologies. According to a clinical study published by NLM in April 2022, 122 patients in the country suffering from idiopathic intracranial hypertension (IIH) were evaluated, out of which headache was the most common symptom among 114 that is 93.4% of the patient pool. Therefore, with such disorders, the country is in huge need of intracranial pressure monitoring devices to minutely identify symptoms and provide treatment solutions.
The market in China is witnessing significant growth opportunities owing to the occurrence of neurological disorders and innovation in technology advancements. According to the February 2023 Frontiers Organization report, a clinical study was conducted on the provision of neuro care in the country. It was found that approximately 96,201 patients suffered from neurological illnesses with an average fatality rate of 4.1%. In addition, the study reported that cerebrovascular disease was the most common form of severe neurological disease with a prevalence rate of 55.2%, thus driving the market demand.
Key Intracranial Pressure Monitoring Devices Market Players:
- Medtronic
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Codman & Shurtleff Inc.
- RAUMEDIC AG
- Vittamed
- Sophysa
- Orsan Medical Technologies
- Integra LifeSciences Holdings Corporation
- Spiegelberg GmbH & Co. KG
- Natus Medical Incorporated
- Gaeltec Devices
- Third Eye Diagnostics
- Vivonics Inc.
- DePuy Synthes
- Luciole Medical AG
- Aesculap, Inc.
With the increasing demand for intracranial pressure monitoring devices, key companies are fast-moving up the manufacturing process while also improving it with cost-effective solutions. In addition, effective strategies such as partnerships, agreements, mergers, and acquisitions are also driving the expansion of the intracranial pressure (ICP) monitoring devices market globally. For instance, in December 2022, Natus Medical Incorporated entered into a decisive agreement to acquire Micromed Holding SAS. With this acquisition, both these companies provided consumers with a best-in-class portfolio of neurodiagnostic and neuromonitoring solutions, thereby driving the demand for intracranial pressure monitoring devices.
Here's the list of some key players:
Recent Developments
- In August 2024, Aesculap, Inc. in partnership with Christoph Miethke GmbH & Co. KG stated that the U.S. FDA granted breakthrough device designation for the M.scio System. This unique device is designed to offer access to long-term monitoring of cerebrospinal fluid for the management of hydrocephalus via a permanent, fully implantable sensor.
- In April 2024, Integra LifeSciences Holdings Corporation declared the successful Q1 2024 U.S. relaunch of its CereLink ICP Monitoring System. This device provides clinicians with uncompromised progressive continuous monitoring with nominal drift, MR conditional ability, durable, flexible sensors, and cutting-edge data presentation features.
- Report ID: 7428
- Published Date: Aug 12, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Intracranial Pressure Monitoring Devices Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




