Computer System Validation Market Outlook:
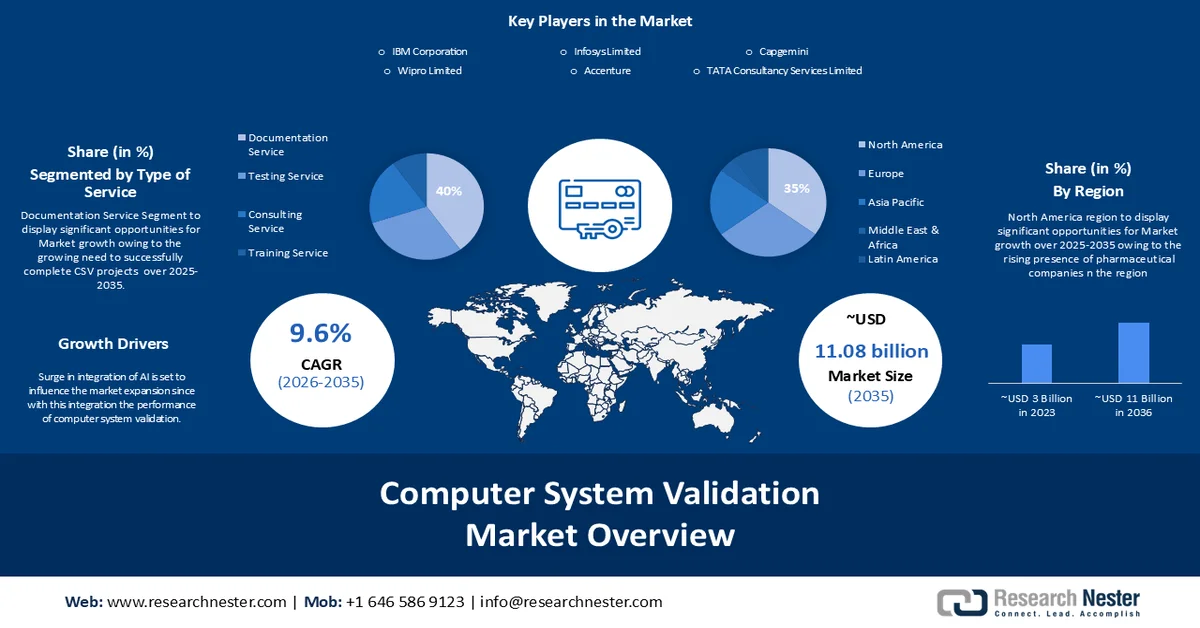
Computer System Validation (CSV) Market size was valued at USD 4.43 billion in 2025 and is set to exceed USD 11.08 billion by 2035, expanding at over 9.6% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of computer system validation is estimated at USD 4.81 billion.
This growth of the computer system validation market is set to be influenced by the growing risk of cyber security. For instance, every day, almost 3999 new cyberattacks take place. A ransomware assault may cause a corporation to suffer catastrophic financial losses about every 13 seconds, and over 559,000 new malware samples are found every day. Hence, organizations need to guarantee the security and integrity of their data, especially in regulated industries where sensitive data is involved. CSV ensures data integrity and protects against security breaches by evaluating data processing, transmission, and storage methods.
Furthermore, Computer systems become increasingly sophisticated as a result of changes in technology and the digital age. Numerous parts, interfaces, and linked procedures could be present in these systems. CSV services are in greater demand since validating such complex systems demands requires particular expertise and expertise.
Key Computer System Validation (CSV) Market Insights Summary:
Regional Insights:
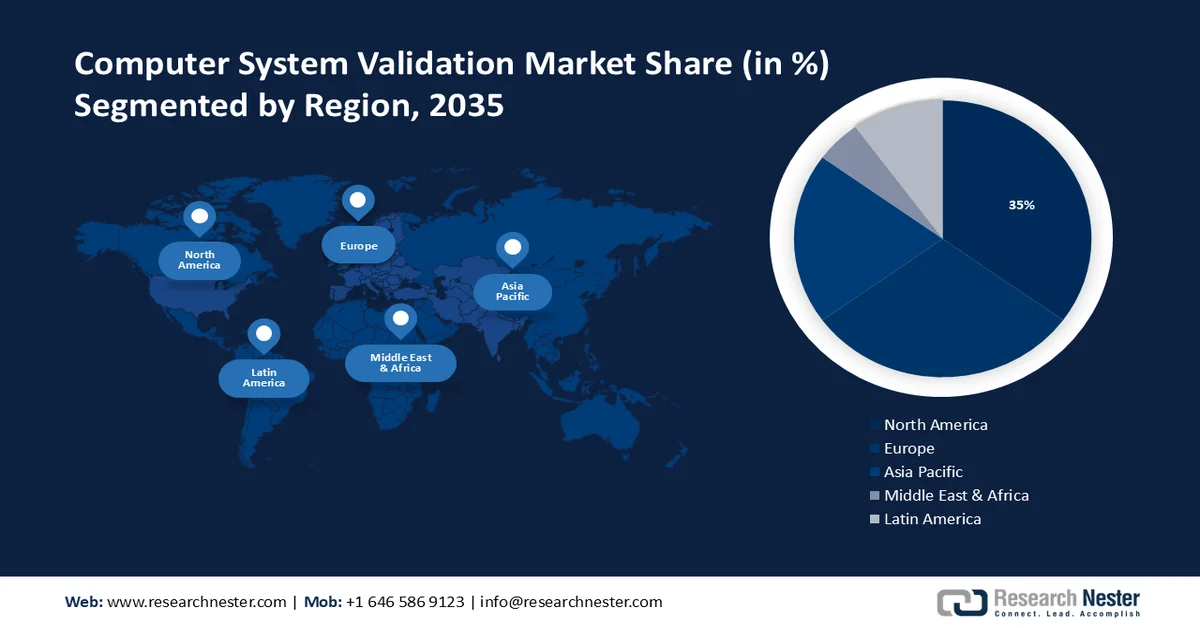
- North America is projected to capture 35% revenue share by 2035 in the computer system validation market, impelled by rising presence of pharmaceutical companies in this region.
- Europe is anticipated to witness notable revenue growth by 2035, propelled by increasing investment in pharma 4.0 and the rising launch of various developments by key players.
Segment Insights:
- The documentation service segment is projected to account for 40% share of the computer system validation market by 2035, driven by the critical need for comprehensive documentation to ensure GxP compliance and consistent system validation.
- The pharmaceuticals segment is expected to hold 40% share of the market by 2035, fueled by stringent regulatory scrutiny and increasing research and development investments within the pharmaceutical industry.
Key Growth Trends:
- Growing Integration of AI & MI in Computer System Validation
- Rising Adoption of Cloud Computing
Major Challenges:
- Growing Cost & Complexity
- Changing Regulation Landscape
Key Players: KAPCI Coating, FENZI S.p.A, BASF SE, CHROMA AUSTRALIA PTY. LTD., Hoechst AG, Hangzhou Han-Color Chemical Co. Ltd., SpecialChem, Anupam Colours & Chemical Industries, Skychem Corporation Limited, Sudarshan Chemical Industries Limited.
Global Computer System Validation (CSV) Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 4.43 billion
- 2026 Market Size: USD 4.81 billion
- Projected Market Size: USD 11.08 billion by 2035
- Growth Forecasts: 9.6% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (35% Share by 2035)
- Fastest Growing Region: Asia Pacific
- Dominating Countries: United States, Germany, Japan, United Kingdom, Canada
- Emerging Countries: India, China, Japan, South Korea, United States
Last updated on : 25 February, 2026
Computer System Validation Market - Growth Drivers and Challenges
Growth Drivers
-
Growing Integration of AI & MI in Computer System Validation- The world of CSV has been completely transformed by artificial intelligence. The way people approach system validations is changing as a result of useful applications that use AI-driven approaches including machine learning, predictive analytics, and automated test execution. These technologies increase validation processes' correctness and dependability in addition to their efficiency. AI is capable of streamlining test cases, quickly spotting possible inconsistencies, and guaranteeing that systems are not only compliant but also performing at the highest possible safety standards.
-
Rising Adoption of Cloud Computing- Cloud computing is becoming more and more popular across all industries. For instance, in 2023, over 89% of organizations were utilizing the cloud across the world. For many businesses that are required to store, process, and access data and apps online, cloud computing is a well-liked and affordable solution. However, in a regulated sector that adheres to good manufacturing practices standards, including biotechnology, pharmaceuticals, or medical devices, one must make sure that the cloud-based systems and applications have been verified in compliance with all applicable rules and regulations. Particularly, to handle the particular difficulties and factors involved in verifying cloud-based systems, such as vendor certification, data residency, and system accessibility, CSV is developing.
-
Surge in Adoption of Computer System Validation by Pharmaceutical Companies- Nowadays, a validation program must be established to show that any procedure, process, equipment, material, activity, or system truly produces the expected results. This requirement applies to the health industries, which include pharmaceuticals (human and veterinary), petrochemicals, (APIs and excipients), biologics, biotechnology, blood products, and medical devices. Validation is necessary to meet normative requirements and ensure the integrity and traceability of information as well as product quality for computerized systems that affect patient health, insurance quality, good practices, and product quality (GxP, for instance, in the case of those that support production processes, input and finished product storage, documentation management, electronic records, and more).
Challenges
-
Growing Cost & Complexity - CSV procedures may be difficult and time-consuming, requiring specialized knowledge, supplies, and paperwork. The complexity and cost of CSV can also rise with the use of complicated computer systems and technologies like artificial intelligence and cloud computing. Implementing CSV can be difficult and more expensive overall because of the requirement for highly qualified staff, validation instruments, and copious amounts of documentation.
-
Changing Regulation Landscape - Regulations and guidelines are frequently updated and changed in areas including healthcare and pharmaceuticals. It can pose a challenge for organizations to stay abreast of constantly changing legislation and guarantee compliance. Complicateries and delays may arise from the necessity to modify CSV procedures to comply with new standards and regulations.
-
Absence of Standardization
Computer System Validation Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Year |
2026-2035 |
|
CAGR |
9.6% |
|
Base Year Market Size (2025) |
USD 4.43 billion |
|
Forecast Year Market Size (2035) |
USD 11.08 billion |
|
Regional Scope |
|
Computer System Validation Market Segmentation:
Type of Service Segment Analysis
The documentation service segment predicted to account for 40% share of the global computer system validation market during the forecast period. In computer system validation, documentation services are the key components that encompass the creation and execution of the necessary documentation for completing a successful CSV project. The process of making sure a computerized system performs as intended, consistently and reliably, is known as system validation. A system's compliance with applicable GxP requirements and its ability to monitor the quality of the data it processes are both demonstrated via validation, which involves the production and upkeep of records.
Application Segment Analysis
The pharmaceuticals segment is predicted to account for 40% share of the global computer system validation (CSV) market during the forecast period. To manufacture pharmaceutical products such as excipients, biotechnology, Active Pharmaceutical Ingredients (APIs), and more., the pharmaceutical business has to develop a validation strategy that outlines how the equipment, methods, raw materials, and procedures will determine the desired outcomes. CSV inspections and audits by the FDA and other regulatory agencies place a strong emphasis on computerized systems utilized in the pharmaceutical business for the storage, management, or transmission of data about regulatory compliance. Any system needed to create, edit, preserve, or send electronic signatures and electronic records falls under this category. For these systems to be secure and operate as intended, a CSV must be performed as part of the validation process. Furthermore, pharmaceutical companies have also observed a rise in investment in research & development which is also boosting the growth of the computer system validation market. Around 243 billion US dollars were spent on research and development in the pharmaceutical sector worldwide in 2022.
Our in-depth analysis of the global computer system validation market includes the following segments:
|
Type of Service |
|
|
Application |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Computer System Validation Market - Regional Analysis
North American Market Insights
North America industry is expected to dominate majority revenue share of 35% by 2035, impelled by rising presence of pharmaceutical companies in this region. This growth is set to be influenced by the rising presence of pharmaceutical companies in this region. As of 2023, the United States is dedicated to more than 4,999 pharmaceutical companies. Moreover, the computer system validation market is shifting towards a lifecycle approach that takes into account a system's whole lifespan in this region. Throughout the whole system lifecycle, from design and development to retirement and decommissioning, organizations are integrating validation processes. Throughout the system's lifetime, this comprehensive approach guarantees continuous compliance, quality, and risk management. Furthermore, these days, new technologies including machine learning and robotic process automation (RPA) are utilized to automate repetitive validation processes, centralize documentation, improve requirement traceability, and expedite workflows for approval and review in this region. This helps companies cut expenses, expedite validation schedules, and enable continuous system validation throughout the system's lifetime.
European Market Insights
The European computer system validation market is also estimated to have notable growth in the revenue over the coming years. This could be owing to the rising launch of various developments by key players in this region. Some of the major contributors to the computer system validation (CSV) market are the United Kingdom, France, and Germany. Moreover, the necessity of secure validation procedures is further highlighted by the growing awareness of cybersecurity dangers, which increases the demand for sophisticated CSV solutions. Additionally, the investment in pharma 4.0 is growing in this region which is also expected to dominate the computer system validation market expansion. CSV used to be a labor-intensive manual process. However, many of the CSV activities can now be automated as a result of Pharma 4.0, which could speed up innovation and increase productivity.
Computer System Validation Market Players:
- Hewlett Packard Enterprise Development LP
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Wipro Limited
- IBM Corporation
- Infosys Limited
- Capgemini
- Cognizant Technology Solutions Corporation
- TATA Consultancy Services Limited
- Deloitte Touche Tohmatsu Limited
- Accenture
- PAREXEL International (MA) Corporation
Recent Developments
- A next-generation compute portfolio that offers a cloud operating experience intended to support hybrid environments and digital transformation was unveiled by Hewlett Packard Enterprise. Organizations can now benefit from intuitive, dependable, and optimized compute resources thanks to the new HPE ProLiant Gen11 servers. These servers are perfect for a variety of contemporary workloads, such as virtualization, analytics, AI, cloud-native apps, machine learning, and graphic-intensive apps.
- To incorporate artificial intelligence (AI) into every platform, tool, and solution used both internally and externally, Wipro Limited, a preeminent provider of technology services and consulting, announced the launch of Wipro ai360, a comprehensive, AI-first innovation ecosystem. This builds on Wipro's ten-year investments in AI. The company said that it would invest $1 billion over the next three years to advance AI capabilities in addition to launching Wipro ai360.
- Report ID: 5839
- Published Date: Feb 25, 2026
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




