Cancer Monoclonal Antibodies Market Outlook:
Cancer Monoclonal Antibodies Market size was over USD 118.59 billion in 2025 and is anticipated to cross USD 589.83 billion by 2035, growing at more than 17.4% CAGR during the forecast period i.e., between 2026-2035. In the year 2026, the industry size of cancer monoclonal antibodies is assessed at USD 137.16 billion.

The market is a rising curve, a trend caused by the synergistic action of well-designed drivers. The incidence of cancer worldwide is on the rise, which demands an ongoing requirement for personalized treatment protocols. For instance, in the February 2024 UICC report, it was updated that, according to the IARC's 2022 report, lung and breast cancer are the most common cancers, accounting for 20 million new cases and 9.7 million deaths worldwide. In addition, regulatory regimes, more streamlined rules for guaranteeing oncology drug approvals, thus enabling patients rapid access to novel treatments.
Furthermore, advancements in technology, especially in production and antibody design, make highly potent and targeted drugs possible. For instance, in July 2024, at the 2024 annual meeting of the American Association for Cancer Research (AACR), researchers notified that the latest manufactured therapeutic cancer vaccine was acknowledged by patients with head and neck squamous cell carcinoma (HNSCC). The new vaccine, TG4050, delivered 30 tailored neoantigens—proteins unique to each patient's tumor—using an engineered version of the vaccinia virus to trigger an antitumor immune response.
Key Cancer Monoclonal Antibodies Market Insights Summary:
Regional Highlights:
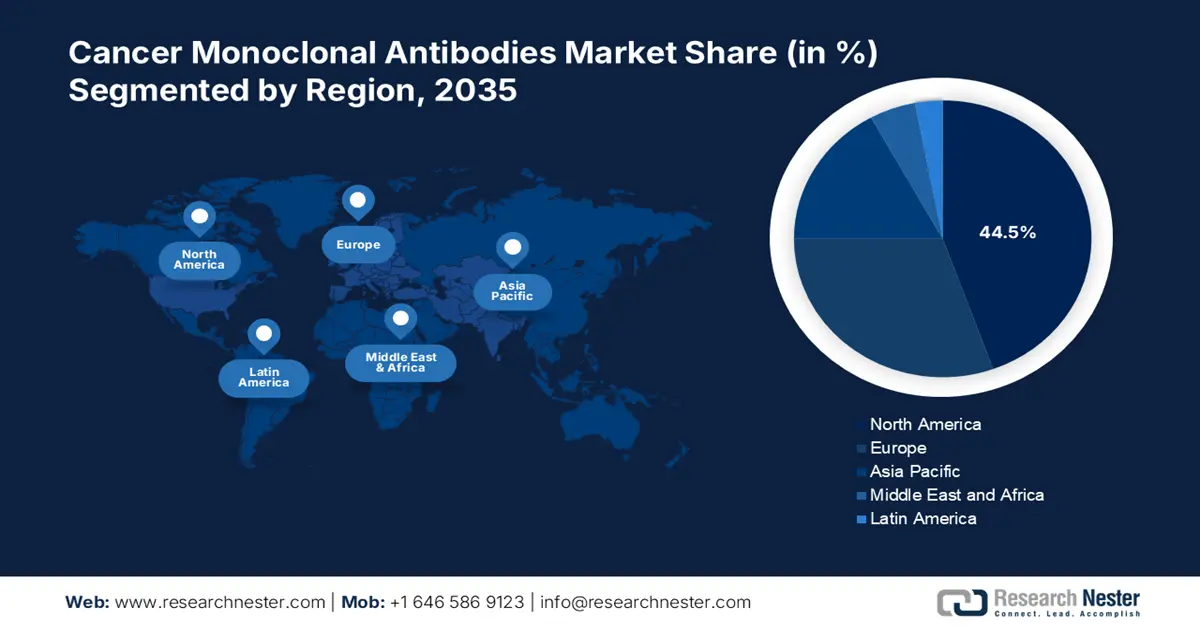
- North America dominates the Cancer Monoclonal Antibodies Market with a 44.5% share, driven by the highly developed healthcare system and rising incidence of cancer, fostering growth through 2035.
Segment Insights:
- The Blood Cancer segment is projected to hold over 29.5% market share by 2035, driven by the efficacy of monoclonal antibodies in hematologic malignancies.
- Trastuzumab (Herceptin) segment is forecasted to secure a lucrative share by 2035, propelled by growing R&D focus on naked monoclonal antibodies and favorable regulatory support.
Key Growth Trends:
- Accelerated biosimilar developments
- Growing understanding of cancer immunology
Major Challenges:
- Manufacturing complexities
- Tumor heterogeneity and resistance
- Key Players: Novartis AG, F. Hoffmann-La Roche Ltd., Bristol Myers Squibb Co., GlaxoSmithKline plc, Johnson & Johnson, AstraZeneca plc, AbbVie, and more.
Global Cancer Monoclonal Antibodies Market Forecast and Regional Outlook:
Market Size & Growth Projections:
- 2025 Market Size: USD 118.59 billion
- 2026 Market Size: USD 137.16 billion
- Projected Market Size: USD 589.83 billion by 2035
- Growth Forecasts: 17.4% CAGR (2026-2035)
Key Regional Dynamics:
- Largest Region: North America (44.5% Share by 2035)
- Fastest Growing Region: Asia-Pacific
- Dominating Countries: United States, Germany, China, Japan, United Kingdom
- Emerging Countries: China, India, Japan, South Korea, Brazil
Last updated on : 12 August, 2025
Cancer Monoclonal Antibodies Market Growth Drivers and Challenges:
Growth Drivers
- Accelerated biosimilar developments: One of the main growth drivers in the cancer monoclonal antibodies market is escalated biosimilar development activities. For instance, in June 2023, a global roadmap to expand patient access to biologic medicines was unveiled by Sandoz. As a component of the Act4Biosimilars initiative, founded and started by the company in 2022, the Act4Biosimilars action plan seeks to raise the adoption of biosimilars globally by at least 30% in over 30 countries by 2030. In addition, incremental competition by way of the introduction of biosimilars promotes innovation and drives prices even lower, thereby enhancing the viability of the market and growing in the long term.
- Growing understanding of cancer immunology: A prominent growth driver for the market is driven by the greater capacity to recognize and target particular tumor-associated antigens and immune checkpoints and thus allow space for more targeted therapy. For instance, in January 2025, researchers from the IIT Guwahati and Bose Institute Kolkata led a multi-institutional study that produced a sophisticated injectable hydrogel for the treatment of localized cancer. The side effects of traditional cancer treatments are greatly decreased by this novel hydrogel-based therapy, which delivers anti-cancer medications straight to tumor sites.
Challenges
- Manufacturing complexities: A built-in barrier to the cancer monoclonal antibodies market is complications that lead to complex scalability, and costly manufacturing. The barrier is most directly relevant to the inherent complexity of biopharmaceutical manufacturing being a highly complex process that requires facilities and high-quality controls. Processes are susceptible to heterogeneity and contamination since manufacturing steps are line-specific and involve intricate purification procedures. Such needs of skilled workers and equipment combined with the possibility of batch failure restrict the capacity for manufacture and increase the final cost of such life-saving treatments.
- Tumor heterogeneity and resistance: The biggest source of the issue is the intrinsic genetic instability of tumor cells generating heterogeneous subpopulations within one tumor. This permits resistant clones to be enriched and amplified even despite the initial efficacy of the targeted therapies. Therefore, the changing tumor cell phenotype with their differential antigen expression and signaling pathways require combination treatments and adaptive treatments to avoid resistance and attain long-term response. Japan's monoclonal antibody cancer culture is propelled by a distinctive combination of demographic transition, technological capacity, and health policy.
Cancer Monoclonal Antibodies Market Size and Forecast:
| Report Attribute | Details |
|---|---|
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
CAGR |
17.4% |
|
Base Year Market Size (2025) |
USD 118.59 billion |
|
Forecast Year Market Size (2035) |
USD 589.83 billion |
|
Regional Scope |
|
Cancer Monoclonal Antibodies Market Segmentation:
Application (Blood Cancer, Breast Cancer, Lung Cancer, Melanoma, Colorectal Cancer, Liver Cancer)
Blood cancer segment is set to hold over 29.5% cancer monoclonal antibodies market share by the end of 2035. The most obvious reason for such extensive prevalence is the accessibility and convenience of the tumor cells and marrow targets to circulation for direct exposure to monoclonal antibodies. For instance, in January 2024, Merck's acquisition of Harpoon Therapeutics along with its phase 1 DLL3 targeting T-cell engager, placed it among the top ten solid M&A deals for cancer. A potential safety benefit of Harpoon's asset in testing for neuroendocrine tumors and small-cell lung cancer is that it only activates when it reaches diseased cells. Therefore, the established efficacy of monoclonal antibodies to achieve long-lasting remissions and increase survival in such hematologic malignancies confirms that they are the treatment mainstay for this category.
Monoclonal Antibody Therapies (Bevacizumab (Avastin), Rituximab (Rituxan), Trastuzumab (Herceptin), Cetuximab (Erbitux), Panitumumab (Vectibix))
Based on the monoclonal antibody therapies segment, the trastuzumab (Herceptin) segment is anticipated to garner a lucrative share by 2035. It is impacted by the expansion of the monoclonal antibody industry as well as a strong focus on the study and creation of therapies based on naked monoclonal antibodies. For complex conditions, they offer extremely effective medication therapy. The development of such innovative therapies is also greatly aided by state support and lax regulatory compliance. For instance, in May 2024, the FDA accelerated approval for trastuzumab deruxtecan, commonly referred to as T-DXd, to treat patients with advanced solid cancer whose tumors are HER2-positive.
Our in-depth analysis of the global market includes the following segments:
|
Application |
|
|
Type |
|
|
Monoclonal Antibody Therapies |
|
|
End user |
|

Vishnu Nair
Head - Global Business DevelopmentCustomize this report to your requirements — connect with our consultant for personalized insights and options.
Cancer Monoclonal Antibodies Market Regional Analysis:
North America Market Statistics
North America cancer monoclonal antibodies market is expected to capture revenue share of over 44.5% by 2035. This growth is attributed to the highly developed healthcare system, the rise in major industry players, the rising incidence of cancer, and the growing knowledge of cutting-edge treatments across the region. Furthermore, the strong presence and expertise of the pharmaceutical and biotechnology industries have become more proficient in discovering the field of cancer monoclonal antibodies, further amplifying the market growth.
The U.S. cancer monoclonal antibodies market is growing exponentially owing to the speedy and successful trials of drugs. For instance, in December 2024, GSK plc declared that the US FDA awarded Jemperli (dostarlimab) breakthrough therapy designation for the treatment of patients with locally advanced mismatch repair deficient (dMMR)/microsatellite instability-high (MSI-H) rectal cancer. To speed up the development and evaluation of medications that have the potential to treat serious conditions, this therapy designation was established once after receiving fast-track designation for the same patient population in January 2023.
The Canada market is likely to witness substantial growth during the forecast period due to the surging instances of cancers across the country. For instance, as per the Canadian Cancer Statistics 2023, it was found that 239,100 new cases of cancer were diagnosed in Canada. In addition, an estimated 31,000 cases of lung cancer were discovered, making it the most frequently diagnosed cancer in Canada followed by breast cancer (29,700), prostate cancer (25,900), and colorectal cancer (24,100). About half (46%) of all cancer diagnoses in 2023 were anticipated to be the four most frequently diagnosed cancers.
Asia Pacific Market Analysis
The Asia Pacific cancer monoclonal antibodies market is predominantly driven by the rising adoption of biosimilars. Moreover, growing interest in genomic research and development, as well as monoclonal antibodies' ability to specifically target cancer spur the market growth. For instance, in January 2025, it was published that the Government of India announced the launch of the Framework for Exchange of Data Protocols (FeED) and the Indian Biological Data Centre (IBDC) Portals, which will produce 10,000 whole genome samples available to researchers worldwide, marking a significant step towards genomics self-reliance. By providing easy access to important genetic data, these will help scientists investigate genetic variations and create more precise genomic tools.
The cancer monoclonal antibodies market in India is witnessing lucrative growth opportunities owing to the regulatory push for novel drugs. For instance, in January 2025, the Association for the Advancement of Blood & Biotherapies unveiled that varnimcabtagene autoleucel (var-cel) is marketed as Qartemi by Immuneel Therapeutics. It is a CD 19-targeted chimeric antigen receptor (CAR) T-cell therapy, approved by India's Central Drugs Standard Control Organization (CDSCO). It is used to treat adult patients with relapsed or refractory B-cell non-Hodgkin'slymphoma. Based on the results of the phase 2 IMAGINE trial, an overall response rate was 83.3% at 90 days among patients who received the treatment.
The market in China is experiencing fast-paced growth owing to the conducive ecosystem for advancements in drugs. For instance, in May 2023, BeiGene announced that the China National Medical Products Administration (NMPA) approved four applications for its Bruton's tyrosine kinase inhibitor (BTKi), BRUKINSA (zanubrutinib). These include two Supplemental New Drug Applications for adults who have not yet received treatment for Waldenström's macroglobulinemia (WM), small lymphocytic lymphoma (SLL), or chronic lymphocytic leukemia (CLL), as well as two supplemental applications for conversions from conditional approval to ordinary approval.
Key Cancer Monoclonal Antibodies Market Players:
- Amgen Inc
- Company Overview
- Business Strategy
- Key Product Offerings
- Financial Performance
- Key Performance Indicators
- Risk Analysis
- Recent Development
- Regional Presence
- SWOT Analysis
- Eli Lilly and Company
- Merck & Co., Inc
- Novartis AG
- F. Hoffmann-La Roche Ltd.
- Bristol Myers Squibb Co.
- GlaxoSmithKline plc
- Johnson & Johnson
- AstraZeneca plc
- AbbVie
The cancer monoclonal antibodies market is characterized to develop therapeutics that align with current trends in the healthcare industry. The pharmaceutical giants enter into strategic moves and discoveries to support the development of novel products and the exploration of new technologies. For instance, in July 2022, Cizzle used Western Blotting (WB) and enzyme-linked immunosorbent assay (ELISA) assays to develop and test a prototype test for early detection of lung cancer based on the CIZ1b biomarker, with high sensitivity and a low false-positive rate that is clinically useful.
Here's the list of some key players:
Recent Developments
- In January 2025, AbbVie and Neomorph, Inc. announced a partnership and option-to-license arrangement to create innovative molecular glue degraders for a variety of immunological and oncological targets.
- In March 2023, Pfizer Inc. and Seagen Inc. announced to have reached a final agreement to acquire Seagen for a total value of USD 43 billion. By fusing the strength of Pfizer's capabilities with the Seagen's antibody-drug conjugate technology, the companies hope to speed up the next generation of cancer discoveries.
- Report ID: 7328
- Published Date: Aug 12, 2025
- Report Format: PDF, PPT
- Explore a preview of key market trends and insights
- Review sample data tables and segment breakdowns
- Experience the quality of our visual data representations
- Evaluate our report structure and research methodology
- Get a glimpse of competitive landscape analysis
- Understand how regional forecasts are presented
- Assess the depth of company profiling and benchmarking
- Preview how actionable insights can support your strategy
Explore real data and analysis
Frequently Asked Questions (FAQ)
Cancer Monoclonal Antibodies Market Report Scope
Free Sample includes current and historical market size, growth trends, regional charts & tables, company profiles, segment-wise forecasts, and more.
Connect with our Expert
Copyright @ 2026 Research Nester. All Rights Reserved.




